Abstract
Idiopathic interstitial pneumonia frequently causes severe pulmonary restriction that in turn makes mechanical ventilation difficult. We report the case of a 44-year-old woman who developed a refractory severe hypercapnic respiratory failure (PaCO2 281 mm Hg, pH 6.77) despite mechanical ventilation with high inspiratory pressure and PEEP. A pumpless extracorporeal lung assist device, Novalung, was used as rescue therapy for carbon dioxide removal, enabling lung-protective ventilation and normalization of life-threatening acidosis. Open lung biopsy revealed an idiopathic interstitial pneumonia with histological features of a nonspecific interstitial pneumonia. Corticosteroid therapy led to progressive improvement of pulmonary function, soon permitting cessation of mechanical ventilation and extracorporeal therapy. The patient was discharged from the intensive care unit after 20 days. This case demonstrates the successful use of pumpless extracorporeal lung assist as an alternative device to pump-driven extracorporeal membrane oxygenation in severe hypercapnic respiratory failure secondary to nonspecific interstitial pneumonia.
Introduction
Mechanical ventilation is a life-saving intervention for patients with acute respiratory distress syndrome (ARDS). However, mechanical ventilation contributes to and perpetuates lung injury, referred to as ventilator-induced lung injury (VILI), by imposing damaging forces of tear and shear. Both VILI and mortality can be reduced by lung-protective ventilation. This strategy consists of low tidal volumes (6 mL/kg predicted body weight), an adequate PEEP, and limiting the inspiratory plateau pressures to a level of 30 cm H2O.1
In life-threatening forms of respiratory failure, unresponsive to conventional invasive mechanical ventilation, pump-driven devices for veno-venous extracorporeal membrane oxygenation (ECMO) have been used as rescue therapy for more than 3 decades.2 The arteriovenous pumpless extracorporeal lung assist (PECLA) device (Novalung, Talheim, Germany), also called an interventional lung assist (iLA), is a device for extracorporeal carbon dioxide removal in patients with severe hypercapnic respiratory failure, facilitating lung-protective ventilation3 (Fig. 1).
PECLA device in situ. In front: gas exchange membrane, with sweep gas entering at the back side (green tube).
The PECLA cannulas are inserted in the femoral artery and vein by means of the Seldinger technique. The driving force through the extracorporeal gas exchange membrane is the arteriovenous pressure gradient. The blood flow through this artificial shunt is usually in the range of 1.0–1.5 L/min, and is dependent on the length and diameter of the cannulas, cardiac output, and mean arterial pressure. The degree of carbon dioxide removal depends not only on the blood flow but also on the gas flow through the membrane (sweep gas). The degree of extracorporeal oxygenation is not clinically relevant. Further details of the PECLA have been described elsewhere.3
Case Report
A 44-year-old female patient presented to a secondary level community hospital with a short history of dyspnea in combination with a non-productive cough and fever. Comorbidities and risk factors were an anxiety disorder treated with quetiapine. Although the patient had a history of smoking (30 pack-years), review of systems revealed no symptoms or signs of chronic lung disease. On history the patient had normal exercise tolerance. Furthermore there was no history of rheumatic disease. Blood tests showed only a minor elevation of inflammatory markers: white blood cells 14.8 × 109 cells/L (standard value 3.8–11.0 × 109 cells/L), C-reactive protein 21.0 mg/L (standard value < 5 mg/L), procalcitonin 0.23 μg/L (standard value < 0.1 μg/L). Chest x-ray showed mild bilateral interstitial infiltrations, and there were no consolidations. The suspected community-acquired pneumonia was initially treated with ceftriaxone and clarithromycin. After 2 weeks of treatment her condition deteriorated, with respiratory failure requiring intermittent noninvasive ventilation. Later chest x-rays showed increasing reticular opacities and small pleural effusion, but no alveolar infiltrations. Antibiotic treatment was changed to piperacillin-sulbactam and moxifloxacin. Within 24 hours the patient required endotracheal intubation and pressure controlled ventilation, with a mandatory respiratory rate of 30 breaths/min and a minute volume of 7.0 L/min. Pulmonary compliance was decreased, and high PEEP (16 cm H2O) and inspiratory pressures (35 cm H2O) were required to generate tidal volumes of about 230 mL (< 3.5 mL/kg predicted body weight) accompanied by severe hypercapnia (PaCO2 281 mm Hg) in conjunction with severe respiratory acidosis (pH 6.77) (Fig. 2). However, oxygenation impairment was comparatively mild (PaO2/FIO2 256 mm Hg).
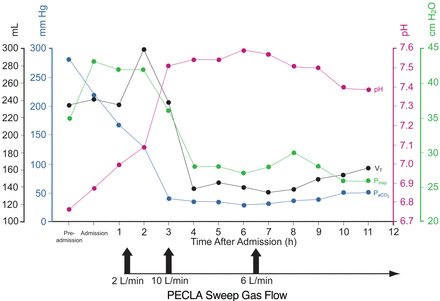
Blood gas analysis and ventilator settings before and up to 12 hours after admission. After implantation of PECLA (left arrow) and flow increase of the sweep gas up to 10 L/min (middle arrow) carbon dioxide removal was sufficient, and PaCO2 and pH normalized. At the same time, lung-protective ventilation settings could be established with low tidal volume (VT) and limited inspiratory pressure (Pinsp). Initial overcompensation (pH 7.54) led to reduction of the gas flow shortly after implantation of PECLA, down to 6 L/min (right arrow).
At this point the patient was transferred to a university hospital. On admission the patient was hemodynamically stable. Low dose vasopressors (4 μg/min norepinephrine) but no positive inotropic agents were administered. Differential diagnoses, including airway occlusion, dislocation of the endotracheal tube, patient-ventilator asynchrony, lung overdistention, and dynamic hyperinflation, were excluded. Neither a clinically important intrinsic PEEP nor relevant air trapping was measured. Pulmonary auscultation indicated diminished air entry bilaterally, with inspiratory fine crackles and no expiratory wheeze. A computed tomography scan of the thorax showed severe bilateral, symmetric interstitial infiltrates, with ground-glass attenuations and accentuated interalveolar and interlobular septal changes, as well as some bronchiectasis in the right lower lobe (Fig. 3).
Computed tomography scan of the chest showing extensive bilateral interstitial infiltrates.
The patient was deeply sedated (Richmond Agitation Sedation Scale of −4) but not medically paralyzed. Because the patient already had bilateral fixed, fully dilated pupils with absence of reaction to light before interhospital transfer, a cerebral computed tomography scan was also performed, but showed no abnormalities. Since no important hypoxemia and hypotension had been reported during the previous clinical course, the pupillary signs were interpreted as an epiphenomenon of hypercapnic encephalopathy and therefore considered potentially reversible.
Changing ventilation modes, altering respiratory rates, lowering the PEEP, and alveolar recruitment maneuvers did not improve ventilation. Because of rapid worsening of the pulmonary restriction (compliance 8.3 mL/cm H2O), with inspiratory pressures of 43 cm H2O and a PaCO2 of 220 mm Hg (see Fig. 2), leading to severe acidosis with signs of encephalopathy, the decision was made to initiate the PECLA as rescue therapy. PECLA cannulas were placed unilaterally in the femoral vessels (arterial cannula 15 French diameter, 90 mm length, venous cannula 17 French diameter, 140 mm length). The hemodynamic status remained stable after initiation of the extracorporeal circulation. Three hours after admission, implantation of PECLA was completed. Lung-protective ventilation was established (tidal volumes of 138 mL, 2 mL/kg predicted body weight) using pressure controlled ventilation with a maximum inspiratory pressure of 28 cm H2O and a PEEP of 10 cm H2O. After an initial and brief overcompensation of the acid-base-status with hypocapnia and alkalosis (PaCO2 29,8 mm Hg, pH 7,59) at a sweep gas flow of 10 L/min, the gas flow of the PECLA was reduced to 6 L/min, resulting in sustained normocapnia (see Fig. 2). Pupillary signs reverted to normal as soon as the hypercapnia had been corrected.
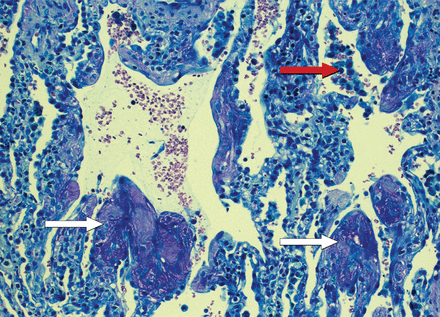
As part of the diagnostic work-up, bronchoscopy showed no macroscopic abnormalities. Neither bronchoalveolar lavage nor cultures from a pleural tap or blood samples revealed positive microbiological results. Further, extensive investigations for viral or bacterial pneumonia, tuberculosis, mycosis, autoimmune disease, or exogenous allergic alveolitis all remained negative. There was no history suggesting drug-induced lung fibrosis. Because the etiology of the interstitial pulmonary infiltrates remained unclear despite extensive diagnostic investigations, an open lung biopsy was performed in the intensive care unit on day 4. The biopsy of the right upper lobe showed diffuse interstitial inflammation and loose interstitial fibrosis, corresponding with a nonspecific interstitial pneumonia (NSIP) with a mixed cellular and fibrosing pattern. There were also areas showing a cryptogenic organizing pneumonia (COP)-like pattern (Fig. 4).
Open lung biopsy with preserved lung architecture. The alveolar septa show mild interstitial chronic inflammation and loose fibrosis (red arrow), additionally, small fibroblastic foci (white arrow) (Giemsa, original magnification ×200).
On the basis of these histological findings, an initial bolus of 100 mg prednisolone was given intravenously, followed by a daily enteral regimen of 0.8 mg prednisolone/kg for a total of 4 weeks. Antibiotic treatment was stopped 10 days after admission. Soon after the initiation of the corticosteroid treatment the patient's pulmonary function progressively improved. A percutaneous dilatational tracheostomy was performed and the patient was weaned from PECLA on day 12 and from invasive mechanical ventilation on day 14. No limb ischemia was observed during the extracorporeal therapy, and Doppler ultrasound did not reveal deep vein thrombosis, arterial injury, or arteriovenous fistula after removal of the PECLA device. On day 18 the tracheal cannula was removed, and on day 20 the patient was transferred to a peripheral ward. At this point she did not require any oxygen supplementation, and the chest x-ray showed no reticular opacities. She was soon discharged home, without any respiratory or neurological sequelae. In a one year follow-up telephone interview the patient appeared intellectually unimpaired, denied any neurological deficits, reported good exercise tolerance (4 flights of stairs without symptoms), and a good quality of life.
Discussion
Idiopathic interstitial pneumonias (IIPs) are a heterogeneous group of diffuse interstitial lung diseases, mostly of unknown etiology. The clinicopathologic consensus classification of IIP from 20024 differentiates 7 entities, which include usual interstitial pneumonia (UIP)/idiopathic pulmonary fibrosis (IPF), acute interstitial pneumonia (AIP, also known as Hamman-Rich syndrome), NSIP, and COP. NSIP occurs mostly in middle-age, non-smoking women. Cardinal symptoms are shortness of breath and non-productive cough. Lung function is limited mainly by restriction, as well as a reduced diffusion capacity. Typical radiological findings are ground glass opacities and patchy alveolar infiltrates. Patients with exogenous allergic alveolitis, connective tissue diseases and drug-induced pulmonary fibrosis may have similar findings.4,5 NSIP is a rare disease and diagnosis is often difficult and requires sufficient clinical, radiological, and histological data.5 In refractory cases of interstitial pneumonias of unclear etiology an open lung biopsy can aid making a diagnosis. The open lung biopsy often leads to changes in management and can safely be performed in selected, mechanically ventilated, critically ill patients.6 The clinical presentation of NSIP can vary from insidious and mild to ARDS. The key histopathologic features are the uniformity of interstitial involvement with a spectrum from a cellular to a fibrosing process (cellular and fibrotic pattern). The histological classification of lung biopsy is very important because there is no recognized clinical distinction between NSIP and UIP/IPF, but NSIP has far better prognosis and treatment options than UIP/IFP, with a better survival associated with the cellular pattern.4,5 In our case the open lung biopsy did not support the diagnosis of UIP or AIP, but verified the diagnosis of NSIP. The recommended first line therapy for NSIP is high dose corticosteroids. In refractory cases these should be combined with azathioprine or cyclophosphamide.7
Survival of patients with severe pulmonary restriction leading to extreme hypercapnia, with PaCO2 values > 280 mm Hg, refractory to invasive mechanical ventilation, have not previously been described. Severe hypercapnia (PaCO2 > 200 mm Hg) usually occurs only in patients who have not been intubated and/or not been adequately ventilated8–10 and can usually be treated by optimizing the airway management and the ventilator settings.
Hypercapnic encephalopathy normally manifests as progressive loss of consciousness and is typically reversible after lowering the PaCO2.11,12 To our knowledge, dilated pupils and absence of pupillary reaction to light, as often seen with hypoxic or hepatic encephalopathy, have not previously been described in severe hypercapnia. Since there was no coincidence with the application or withdrawal of drugs that are known to potentially influence the pupillary motor system (eg, anticholinergic, ß-adrenergic receptor agonistic, or paralytic drugs) and pupillary signs were promptly and fully reversible with normocapnia, we postulate that these signs were an epiphenomenon of the severe hypercapnia.
PECLA has been used in hypercapnic respiratory failure due to ARDS,3,13 asthma,14 and in patients with ARDS and brain injury.15 One study has described a 10-year experience of PECLA in 178 patients from Germany.13 In another retrospective study, PECLA was used in patients with an average PaCO2 value of 60 mm Hg (range 48–80).3 PECLA enables low tidal volume ventilation without acidosis, by removing excess carbon dioxide that is not expired.3 Furthermore, avoiding additional VILI1 allows the lung to heal. It is recommended to increase the PECLA sweep gas-flow by only 1 L/min every 20 min, with readjustment of the respirator setting according to frequently drawn arterial blood gas analysis. This report demonstrates the risk of a potentially harmful metabolic/respiratory overcompensation if sweep gas flow is increased too fast.
Patients with IIP often present with severe pulmonary restriction causing hypercapnic respiratory failure.4,5 Few case reports have been published describing critical clinical courses of IIP causing difficult invasive mechanical ventilation16–18 or requiring ECMO.17 The application of PECLA in a patient with NSIP has not been described until now. Growing evidence suggests that moderate hypercapnic acidosis is well tolerated (permissive hypercapnia) and may even be beneficial as long as lung-protective ventilation is possible.19
There is no evidence of significant survival benefit of PECLA, compared to veno-venous ECMO in hypercapnic respiratory failure, because of lack of prospective trials. However, from the technical and pathophysiological point of view, some major disadvantages of ECMO (high dose heparin therapy, risk of bleeding, mechanical stress on cellular blood components due to the blood pump, activation of coagulation, and high costs3,15) can be avoided with a PECLA. The PECLA surface is heparin-coated. High dose heparin is not obligatory in patients with high risk of bleeding.
Nevertheless, ECMO is sometimes used as a rescue therapy in hypercapnic respiratory failure refractory to conventional mechanical ventilation. This case demonstrates the successful use of a PECLA as an alternative device to pump-driven ECMO in a patient with NSIP. Considering the use of PECLA should be limited to conditions where the underlying pulmonary pathology is treatable and potentially reversible.
Footnotes
- Correspondence: Martin Petzoldt MD, Center of Anesthesiology and Intensive Care Medicine, University Medical Center Hamburg-Eppendorf, Martinistrasse 52, 20246, Hamburg, Germany. E-mail: m.petzoldt{at}uke.de.
Dr Kluge has disclosed a relationship with Novalung.
The other authors have disclosed no conflicts of interest.
- © 2012 by Daedalus Enterprises Inc.