Abstract
Objective
To assess the hysteresis of the pressure–volume curve (PV curve) as to estimate, easily and at the bedside, the recruitability of the lung in ARDS patients.
Design
Prospective study.
Setting
Twelve medico-surgical ICU beds of a general hospital.
Patients
Twenty-six patients within the first 24 h from meeting ARDS criteria.
Intervention
A Quasi-static inflation and deflation PV curve from 0 to 40 cmH2O and a 40 cmH2O recruitment maneuver (RM) maintained for 10 s were successively done with an interval of 30 min in between.
Recordings and calculation
Hysteresis of the PV curve (H PV) was calculated as the ratio of the area enclosed by the pressure volume loop divided by the predicted body weight (PBW). The volume increase during the RM (V RM) was measured by integration of the flow required to maintain the pressure at 40 cmH2O and divided by PBW, as an estimation of the volume recruited during the RM.
Results
A positive linear correlation was found between H PV and V RM (r = 0.81, P < 0.0001).
Conclusions
The results suggest using the hysteresis of the PV curve to assess the recruitability of the lung.
Similar content being viewed by others
Introduction
In the acute respiratory distress syndrome (ARDS), positive end-expiratory pressure (PEEP) may decrease ventilator-induced lung injury by keeping lung regions open that otherwise would collapse [1, 2]. However, the ability of the lung regions to become and remain open, that is “recruitability”, is a prerequisite for a rational determination of the levels of PEEP to be applied [3]. In a large group of ARDS patients, based on CT scan examination, Gattinoni and co-workers [3] found that the recruitability of the lungs varied widely among ARDS patients. Moreover, the effect of PEEP on lung recruitment was closely associated with the recruitability of the lungs estimated from the CT scan. At the same time, the recruitability was weakly estimated at the bedside by PEEP induced changes in physiologic respiratory variables (PaO2/FiO2, PaCO2 or static compliance). In this context, a simple and reproducible bedside method to estimate lungs recruitability is still lacking.
Studies in ARDS patients have suggested that the shape of the inflation limb of the PV curve could predict recruitability [4, 5]. In particular, a lower inflection point and a higher linear compliance (C LIN) have been associated with a large recruitment induced by PEEP [4, 5]. The hysteresis of the pressure–volume curve (PV curve) was mentioned first by Mead [6] in 1957 and was defined as the surface between the inflation and the deflation limb of the quasi-static PV curve. Though the underlying mechanism of such phenomena is not yet fully understood, it has been suggested from mathematical models, [7, 8] animal studies [9, 10] and human observations [11, 12] that the larger the hysteresis, the higher the recruitability of the lung. Interestingly the results presented from the Vieillard–Baron’s study, [5] display an obvious visual difference in the hysteresis between recruiters and non-recruiters.
This preliminary and exploratory study was designed to assess the hysteresis of the PV curve as to estimate, easily and at the bedside, the recruitability of the lung in ARDS patients. To do so, Hysteresis of the quasi static PV curve (H PV) was compared with the volume increase during a recruitment maneuver (V RM).
Materials and methods
Patients
This prospective study was conducted from February 2006 till June 2007 in the 12-beds adult mix-ICU of Font Pré Hospital in Toulon (France). The ethic committee approved the protocol and informed consent was obtained from each patient’s next of kin. Patients were included if they presented early onset (≤24 h) ARDS as defined by the American-European Consensus Conference [13]. Non-inclusion criteria were morbid obesity, chest wall stiffness, moribund status, hemodynamic disorder requiring more than 1.4 μg/Kg/min of epinephrine or norepinephrine, hypovolemia as reflected by a variation in pulse pressure (ΔPP) over 13% [14], increased intracranial pressure, pulmonary emphysema, severe chronic respiratory disease requiring long-term oxygen therapy or long-term mechanical ventilation, or a chest tube in place.
Patients were orally intubated and mechanically ventilated using a Galileo Gold ventilator (HAMILTON MEDICAL AG, Rhäzüns, Switzerland), in adaptive support ventilation (ASV) mode [15]. Settings were adjusted to keep tidal volume (V T) below 10 mL/kg of predicted body weight with a plateau pressure below 30 cmH2O [16]. FiO2 was adjusted to keep SpO2 >90% throughout the study and for the research purpose, PEEP was set at 10 cmH2O for all the patients. Patients were sedated using midazolam and fentanyl to reach a Ramsay score of 6, and paralyzed with a single injection of cisatracurium. Electrocardiogram, blood pressure and pulse oxymetry were monitored throughout the study. PV curve and RM were interrupted in case of bradycardia (≤50/min), hypotension (decrease in systolic blood pressure ≥20 mmHg) or desaturation (decrease in SpO2 ≥5%).
Measurements
Airway pressure and flow were measured using the proximal pneumotachograph of the ventilator (single-use flow sensor, PN 279331, Hamilton Medical, Bonaduz, Switzerland, linear between −120 and 120 L/min with a ±5% error of measure) inserted between the endotracheal tube and the Y-piece. The signal was acquired at 67 Hz and downloaded from the ventilator using specific acquisition software (Datalogger 3.27, Hamilton Medical AG, Rhäzüns, Switzerland). Volume was obtained by integration of the flow signal. Static compliance (Cstat) was measured by the least square fit method over the full respiratory cycle during the basal ventilation period [17]. Plateau pressure and total PEEP were measured using a 5-s end-inspiratory and end-expiratory occlusion, respectively.
Experimental protocol
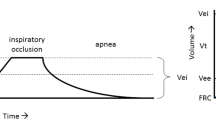
A Quasi-static inflation and deflation PV curve and RM were successively done with an interval of 30 min in between to restore volume and pressure history (Fig. 1).
PV curve
The airway was suctioned and the cuff of the endotracheal tube was controlled to avoid any leak. All equipment connections were also revised to avoid an air leak, which, in addition was verified by observing no changes in airway pressure during a 10-s end-inspiratory pause. Inflation and deflation pressure–volume curves were obtained from 0 to 40 cmH2O using an automatic pressure ramp method (PV tool 2 Hamilton Medical, AG, Bonaduz, Switzerland). The pressure ramp was set at 2.0 cmH2O/s. Resistive pressures were considered negligible by checking the flow-pressure curves showing flow values below 10 L/min during the entire maneuver [18, 19].
Recruitment maneuver
Once volume and pressure history has returned to basal conditions, a single RM consisting of a sustained inflation at 40 cmH2O of airway pressure was performed. Since no consensus exist about the way to perform a RM and because of the concerns regarding safety [20, 21], 10 s duration and a progressive raise in pressure (2 cmH2O/s) were adopted [22]. Using such a method, the volume increase (if any) may be underestimated as compared to the volume increase by a prolonged RM.
Calculations
From the pressure and volume information, the PV curves were drawn using graphic software (Sigma Plot 8.0. SPSS Inc, Chicago, IL USA). Inflation and deflation volume data were corrected for changes in temperature, humidity and oxygen consumption [23, 24]. Loss of volume caused by oxygen consumption was assumed to be 92 mL/min, the average rate of loss in thoracic gas volume measured in patients by Dall’ava-Santucci and colleagues [23]. Because hysteresis depends on the size of the lung [25], H PV was calculated as the ratio of the area enclosed by the pressure volume loop between 0 and 40 cmH2O, measured by planimetry and divided by the predicted body weight (PBW) (Fig. 1). The lower inflection point (LIP), the upper inflection point (UIP) and point of maximum curvature (PMC) of the quasi-static PV curve were determined using build in adjustable cursors to help reduce the interobserver variability in defining the points of interest [26]. In short, a line is adjusted between the two cursors along the inflation and the deflation limbs of the PV curve. The LIP and UIP were defined as the points where the line between the two cursors diverged from the inflation limb and the PMC as the point where the line between the two cursors diverge from the upper linear part of the deflation limb [27] (Fig. 1). The linear compliance (C LIN) was calculated from the linear segment between the LIP and UIP [4, 28] (Fig. 1).
The volume increase during the RM was estimated by the integration of the flow required to maintain the pressure at 40 cmH2O assuming that in leak-free condition, the additional volume needed to maintain the pressure, is an estimation of the volume recruited (Fig. 1). Leaks were ruled out by no change in airway pressure during the 10-s sustained inflation. In case of leaks, we assumed a constant flow with consequently a linear increase in volume without plateau, whereas in the absence of leaks we assumed a volume increase with exponential shape and a plateau. If leaks were suspected, data were rejected from analysis. The volume increase during the RM was as well corrected for temperature, humidity and oxygen consumption [23]. V RM was computed as the ratio between the volume increase during the RM and PBW.
Statistical methods
Statistics were performed using SigmaStat (Version 3.0, SPSS, Inc., Chicago, IL USA). Data are reported as medians (25th–75th quartiles). Linear regressions were performed using Spearman test (Sigma Plot Version 8.0. SPSS Inc., Chicago, IL USA).
Results
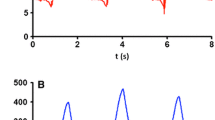
Thirty-four patients were selected, six patients were not included because of exclusion criteria and two patients were rejected for leaks during the RM. The results concern the remaining 26 patients [age = 65 (54–76) years, SAPS II = 57 (39–67), lung injury Score = 3.0 (2.8–3.3)]. Twenty-two patients had pulmonary ARDS (11 pneumonia, 10 inhalation, and 1 chest trauma) and 4 extra-pulmonary ARDS (2 septic shocks, 2 massive transfusions). The respiratory variables at inclusion are described in Table 1. PV and RM curves from typical patients with low hysteresis and a patient with high hysteresis are shown in Fig. 2.
PV curves and RM were not interrupted according to the criteria defined in “Materials and methods” and no pneumothorax was observed after the experiment. The inflation and deflation flow during the maneuvers were 7.1 (5.7–8.2) and 7.6 (5.8–9.0) L/min, respectively.
The LIP, UIP and PMC were 8 (6–10), 29 (27–31) and 24 (21–26) cmH2O, respectively. C LIN was 47 (39–59) mL/cmH2O. A positive linear correlation (r = 0.65, p = 0,001) was found between C LIN and V RM. A positive linear correlation (r = 0.81, P < 0.0001) was also found between H PV and V RM (Fig. 3).
Discussion
This preliminary study reports a significant relationship between hysteresis of the PV curve (H PV) and the volume increase during a RM (V RM), which if confirmed, may help screening at the bedside patients whose lung may benefit from a recruitment maneuver.
The gold standard method to measure recruitability of the lung is CT scan [29, 30]. CT scan detects lung collapse and measures the percentage of volume recruited by a RM [31]. Unfortunately, CT scan is not available at the bedside and moving patients with acute illness to the CT scan may be harmful [32]. Electrical impedance tomography (EIT) is a method for monitoring regional lung function and estimating alveolar recruitment [33, 34]. EIT is not yet available on the market and is mainly used for research purpose. Gas-exchange data would have been helpful though, some sort of dissociation between anatomical recruitment (measure at the CT scan) and functional recruitment (measure by improvement in gas exchanges) has been reported recently [3]. Therefore, a simple and reproducible bedside method to estimate lungs recruitability is still lacking. In the present study, recrutability of the lung was estimated through the volume required to maintain the pressure during a 10-s sustained inflation at 40 cmH2O. This method has not been validated versus CT scan or EIT indeed. Not only such a volume could be recruitment but also leaks, oxygen consumption, viscoelasticity or ventilation inhomogeneity (pendelluft). Leaks were carefully avoided by securing the ventilation circuit, over inflating the endotracheal tube’s cuff and by checking the pressure and volume pattern over time during the sustained inflation (linear increase without plateau in case of leak, exponential increase with a plateau in case of recruitment). Though not measured during the maneuver, oxygen consumption was subtracted from the volume increase according to standard formula based on measurement in similar patients [23]. Finally and considering the RM used (slow pressure raise), the viscoelastic component is probably very limited, as well as the Pendelluft effect.
Though a confirmative CT scan analysis is definitely required; V RM is plausibly the volume recruiting alveoli with high opening pressure and/or long time constant [7]. In the present study a “conservative” RM maneuver was used (progressive increase in pressure and 10 s duration), and because recruitment occurred all along the inflation, V RM probably underestimated the total volume recruited [7].
Hysteresis of the PV curve results from stress relaxation and gas consumption and mainly from recruitment/derecruitment and surface forces [27]. In ARDS patients, hysteresis decreased when PEEP was increased as a result of smaller percentage of lung collapse at the beginning of the PV curve [35]. Moreover, static PV curves show minimal hysteresis under conditions when lung closure and reopening does not occur, either in healthy [36–38] or in diseased lungs [39]. It is therefore reasonable to assume H PV as an estimate of lungs recruitability: the larger the H PV, the more is recruited during an RM. In this study, the role of chest wall was not considered. Airway pressure was standardized to compute H PV, which may result in different transpulmonary pressure according to the chest wall compliance. Patients with supposed stiff chest wall (obesity and chest wall stiffness) were not included and though hysteresis seems similar in animal’s excised lung when inflated at the same volume as in intact animal [40], the effect of chest wall on the hysteresis should be further investigated.
C LIN has been suggested to predict recrutability [4, 5]. In this study, correlation between C LIN and V RM was significant though moderate (r = 0.65), as compared to the correlation found between H PV and V RM (r = 0.81). C LIN is a measure of the recruitment that occurs during a definite part of inflation, that is, between the points of inflection (which suppose some inflections on the curve), while H PV depends on inflation and deflation over the entire range of pressure and does not depend on how the linear compliance is measured. Both indexes predict recrutability but H PV may be better correlated with the volume recruited.
Gattinoni et al. [3] demonstrated that knowledge of the percentage of potentially recruitable lung (recruitability) may be important for establishing the therapeutic efficacy of PEEP. Setting PEEP without information on the recruitability of the lung and without previous RM may offset the possible benefits of PEEP. At the same time, recruitability is weakly estimated at the bedside by PEEP-induced changes in physiologic respiratory variables (PaO2/FiO2, PaCO2 or static compliance) [3]. CT scan might help to assess alveolar recruitment and lung overinflation but is yet difficult to integrate into clinical practice. In this context, the present results suggest that hysteresis may help to estimate lung’s recruitability at the bedside. As simple “PV tools” are now built in commercial ventilators, H PV might be easily available at the bedside [41, 42]. In the present study, H PV has been calculated as the ratio of the area enclosed by the pressure volume loop between 0 and 40 cmH2O, measured by planimetry and divided by PBW. A simpler approach could be used to assess H PV, such as the volume difference between the deflation and the inflation limb of the PV curve at a given airway pressure [27]; more complex approach can also be used, such as the transpulmonary instead of the transthoracic pressure specially in patients with low chest wall compliance [43]. Further studies should define the best approach in term of easiness and reproducibility at the bedside. In certain patients, RM may be poorly tolerated (hemodynamic instability, pulmonary emphysema…) and RM is still arguable in terms of safety [20]. In these situations, an index of recruitability, such as the hysteresis which could be easily automatized, would certainly help.
In conclusion, the present study suggests that the hysteresis of the PV curve might help to assess the recruitability of the lung, and define which patient needs recruitment maneuver before setting PEEP.
References
Crotti S, Mascheroni D, Caironi P, Pelosi P, Ronzoni G, Mondino M, Marini J, Gattinoni L (2002) Recruitment and de-recruitment during acute respiratory failure. A clinical study. Am J Respir Crit Care Med 164:131–140
Barbas V, De Matos J, Pincelli P, Borges R, Antunes T, Monteiro de Barros J, Okamoto V, Borges JB, Amato M, De Carvalho C (2005) Mechanical ventilation in acute respiratory failure: recruitment and high positive end-expiratory pressure are necessary. Curr Opin Crit Care 11:18–28
Gattinoni L, Caironi P, Cressoni M, Chiumello D, Ranieri M, Quintel M, Russo S, Patroniti N, Cornejo R, Bugedo G (2006) Lung recruitment in patients with the Acute Respiratory Distress Syndrome. N Engl J Med 354:1775–1786
Maggiore SM, Jonson B, Richard JC, Jaber S, Lemaire F, Brochard L (2001) Alveolar derecruitment at decremental positive end-expiratory pressure levels in acute lung injury. Am J Respir Crit Care Med 164:795–801
Vieillard-Baron A, Prin S, Chergui K, Page B, Beauchet A, Jardin F (2003) Early patterns of static pressure-volume loops in ARDS and their relations with PEEP-induced recruitment. Intensive Care Med 29:1929–1935
Mead J, Whittenberger JL, Radford EP (1957) Surface tension as a factor in pulmonary volume-pressure hysteresis. J Appl Physiol I0:191–196
Hickling K (1998) Recruitment greatly alters the pressure volume curve: a mathematical model of ARDS lungs. Am J Respir Crit Care Med 158:194–202
Hickling KG (2001) Best compliance during a decremental, but not incremental, positive end-expiratory pressure trial is related to open-lung positive end expiratory pressure: a mathematical model of acute respiratory distress syndrome lungs. Am J Respir Crit Care Med 163:69–78
Rimensberger PC, Pristine G, Mullen BM, Cox PN, Slutsky AS (1999) Lung recruitment during small tidal volume ventilation allows minimal positive end-expiratory pressure without augmenting lung injury. Crit Care Med 27:1940–1945
Henzler D, Hochhausen N, Dembinski R, Orfao S, Rossaint R, Kuhlen R (2007) Parameters derived from the pulmonary pressure-volume curve, but not the pressure-time curve indicate recruitment in experimental lung injury. Anesth Analg 105:1072–1078
Richard JC, Brochard L, Vandelet P, Breton L, Maggiore SM, Jonson B, Clabault K, Leroy J, Bonmarchand G (2003) Respective effects of end-expiratory and end-inspiratory pressures on alveolar recruitment in acute lung injury. Crit Care Med 31:89–92
Matamis D, Lemaire F, Harf A, Brun-Buisson C, Ansquer JC, Atlan G (1984) Total respiratory pressure-volume curves in the adult respiratory distress syndrome. Chest 86:58–66
Bernard GR, Artigas A, Brigham KL, Carlet J, Falke K, Hudson L, Lamy M, Le Gall JR, Morris A, Spragg R (1994) Report of the American-European Consensus Conference on acute respiratory distress syndrome: definitions, mechanism, relevant outcomes, and clinical trial coordination. Consensus Committee. Am J Respir Crit Care Med 149:818–824
Michard F, Boussat S, Chemla D, Anguel N, Mercat A, Lecarpentier Y, Richard C, Pinsky M, Teboul JL (2000) Relation between respiratory changes in arterial pulse pressure and fluid responsiveness in septic patients with acute circulatory failure. Am J Respir Crit Care Med 162:134–138
Arnal JM, Wysocki M, Nafati C, Donati SY, Granier I, Corno G, Durand-Gasselin J (2008) Automatic selection of breathing pattern using adaptive support ventilation. Intensive Care Med 34:75–81
Arnal JM, Wysocki M, Garcin F, Donati SY, Granier I, Durand-Gasselin J (2007) Adaptive Support Ventilation (ASV®) automatically adapts a protective ventilation in ARDS patients. Am J Resp Crit Care Med 175:A244
Iotti GA, Braschi A, Brunner JX, Smits T, Olivei M, Palo A, Veronesi R (1995) Respiratory mechanics by least squares fitting in mechanically ventilated patients: applications during paralysis and during pressure support ventilation. Intensive Care Med 21:406–413
Servillo G, Svantesson C, Beydon L, Roupie E, Brochard L, Lemaire F, Jonson B (1997) Pressure-volume curves in acute respiratory failure: automated low flow inflation versus occlusion. Am J Respir Crit Care Med 155:1629–1636
Lu Q, Vieira SR, Richecoeur J, Puybasset L, Kalfon P, Coriat P, Rouby JJ (1999) A simple automated method for measuring pressure–volume curves during mechanical ventilation. Am J Respir Crit Care Med 159:275–282
Kacmarek R, Kallet R (2007) Should recruitment maneuvers be used in the management of ALI and ARDS? Respir Care 52:622–631
Mols G, Priebe HJ, Guttmann J (2006) Alveolar recruitment in acute lung injury. Br J Anaesth 96:156–66
Odenstedt H, Lindgren S, Olegard C, Erlandsson K, Lethvall S, Aneman A, Stenquist O, Lundin S (2005) Slow moderate pressure recruitment maneuver minimizes negative circulatory and lung mechanic side effects: evaluation of recruitment maneuver using electric impedance tomography. Intensive Care Med 31:1706–1714
Dall’ava-Santucci J, Armaganidis A, Brunet F, Dhainaut JF, Chelucci GL, Monsallier JF, Lockhart A (1988) Causes of error of respiratory pressure-volume curves in paralyzed subjects. J Appl Physiol 64(1):42–49
Gattinoni L, Mascheroni D, Basilico E, Foti G, Pesenti A, Avalli L (1987) Volume/pressure curve of total respiratory system in paralysed patients: artefacts and correction factors. Intensive Care Med 13:19–25
Bachofen H, Hildebrandt J (1971) Area analysis of pressure volume hysteresis in mammalian lungs. J Appl Physiol 30:493–497
Mehta S, Stewart TE, MacDonald R, Hallett D, Banayan D, Lapinsky S, Slutsky A (2003) Temporal change, reproducibility, and interobserver variability in pressure-volume curves in adults with acute lung injury and acute respiratory distress syndrome. Crit Care Med 31(8):2118–2125
Harris RS (2005) Pressure–volume curves of the respiratory system. Respir Care 50(1):78–98
Jonson B, Richard JC, Strauss C, Mancebo J, Lemaire F, Brochard L (1999) Pressure–volume curves and compliance in acute lung injury. Evidence of recruitment above the lower inflexion point. Am J Respir Crit Care Med 159:1172–1178
Gattinoni L, Caironi P, Pelosi P, Goodman LR (2001) What has computed tomography taught us about the acute respiratory distress syndrome? Am J Respir Crit Care Med 164:1701–1711
Malbouisson LM, Muller JC, Constantin JM, Puybasset L, Rouby JJ and CT Study Scan ARDS Group (2001) Computed tomography assessment of positive endexpiratory pressure-induced alveolar recruitment in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med 163:1444–1450
Tokics L, Hedenstierna G, Svensson L, Brismar B, Cederlund T, Lundquist H, Strandgerd A (1996) V/Q distribution and correlation to atelectasis in anesthetized paralyzed humans. J Appl Physiol 81:1822–1833
Waydhas C (1999) Intrahospital transport of critically ill patients. Crit Care 3:R83–R89
Frerichs I, Schiffmann H, Hahn G, Hellige G (2001) Non-invasive radiation- free monitoring of regional lung ventilation in critically ill infants. Intensive Care Med 27:1385–1394
Frerichs I, Dargaville P, Dudykevych T, Rimensberger P (2003) Electrical impedance tomography: a method for monitoring regional lung aeration and tidal volume distribution? Intensive Care Med 29:2312–2316
Dall’ava-Santucci J, Armaganidis A, Brunet F, Dhainaut JF, Nouira S, Morisseau D, Lockhart A (1990) Mechanical effects of PEEP in patients with adult respiratory distress syndrome. J Appl Physiol 68:843–848
Jonson B, Beydon L, Brauer K, Mansson C, Valind S, Grytzell H (1993) Mechanics of respiratory system in healthy anesthetized humans with emphasis on viscoelastic properties. J Appl Physiol 75:132–140
Svantesson C, John J, Taskar V, Evander E, Jonson B (1996) Respiratory mechanics in rabbits ventilated with different tidal volumes. Respir Physiol 106:307–316
Bitzen U, Drefeldt B, Niklason L, Jonson B (2004) Dynamic elastic pressure volume loops in healthy pigs recorded with inspiratory and expiratory sinusoidal flow modulation. Relationship to static pressure-volume loops. Intensive Care Med 30:481–488
Beydon L, Svantesson C, Brauer K, Lemaire F, Jonson B (1996) Respiratory mechanics in patients ventilated for critical lung disease. Eur Respir J 9:262–273
Radford EP Jr. Static mechanical properties of mammalian lungs. In: Fenn WO, editor. Handbook of physiology. Bethesda: American Physiological Society; 1964–1965:429–449
Jonson B (2005) Elastic pressure-volume curves in acute lung injury and acute respiratory distress syndrome. Intensive Care Med 31:205–212
Lucangelu U, Bernabè F, Blanch L (2007) Lung mechanics at the bedside: make it simple. Curr Opin Crit Care 13:64–72
Talmor D, Sarge T, O’Donnell CR, Ritz R, Malhotra A, Lisbon A, Loring SH (2006) Esophageal and transpulmonary pressures in acute respiratory failure. Crit Care Med 34:1389–1394
Conflict of interest
D.D. was supported by Hamilton Medical in presenting the results of this study at international congress. M.W. is an employee of Hamilton Medical and as the Head of Medical Research was involved in the initial discussions regarding the design of the study and in helping to write the manuscript. He was not involved in collecting and analyzing the data.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was conducted in the Intensive Care Unit of Font Pré Hospital, Toulon, France.
Rights and permissions
About this article
Cite this article
Demory, D., Arnal, JM., Wysocki, M. et al. Recruitability of the lung estimated by the pressure volume curve hysteresis in ARDS patients. Intensive Care Med 34, 2019–2025 (2008). https://doi.org/10.1007/s00134-008-1167-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-008-1167-8