Abstract
Evidence-based medicine has assumed a major place in establishing the standard of care for many diseases. Yet practices based largely on clinical experience have often been difficult to change, even when clearly associated with patient harm. In this paper, based on the 27th Philip Kittredge Memorial Lecture, we highlight respiratory care practices that have clearly failed the tests of time and evidence yet sometimes tenaciously persist. We also discuss the appropriate use and potential abuse of evidence-based medicine.
- evidence-based medicine
- asthma
- inhalers
- croup tents
- nebulizers
- oxygen therapy
- continuous positive airway pressure
- intermittent positive pressure breathing
- mucolytics
- dornase alfa
- chronic obstructive pulmonary disease
- sedation
- wheezing
- acetylcysteine
- blow-by
- tracheomalacia
- cough
- bronchodilator
- levalbuterol
- corticosteroids
- myocardial infarction
- meta-analysis
- statistical power
Nothing is more obstinate than a fashionable consensus
—Margaret Thatcher
Introduction
Evidence-based medicine (EBM) has assumed a major place in establishing the standard of care for many diseases. Yet practices based largely on clinical experience have often been difficult to change, even when clearly associated with futility or patient harm. The term “eminence-based medicine” was coined by Isaacs and Fitzgerald in 1999, as a tongue-in-cheek look at EBM.1 They defined eminence-based medicine as, “The more senior the colleague, the less importance they place on the need for anything as mundane as evidence. They have a touching faith in clinical experience defined as making the same mistakes with increasing confidence over an impressive number of years.”
Evidence grading and meta-analyses are used to evaluate the quality and applicability of published studies, and the principles of EBM can be used to help design good studies. As a corollary, poorly designed studies can be used to support dogma and myth.
Dogma is a point or view that is proclaimed with authority, often taught using the adverbs “always” and “never” (Table 1). Because of its authoritative nature, dogma is not subject to questioning. Dogma is often defended by statements such as: “Why do research? If something seems to be working well for our patients and it doesn't appear to be hurting them, why change?” “This is the way we always have done it, and the patients do just fine.” “Who needs data? Common sense is enough!” and “If it ain't broke, don't fix it.”
Definitions
In this paper, based on the 27th Philip Kittredge Memorial Lecture, we highlight respiratory care practices that have clearly failed the tests of time and evidence yet sometimes tenaciously persist. We also discuss the appropriate use and potential abuse of EBM.
Great Myths, Misunderstandings, and Dogma
There have been many examples in pulmonary medicine and respiratory care of dogma perpetuating poor care for our patients. We begin by demonstrating that even great minds can make great mistakes. To quote Nikki Giovanni, “Mistakes are a fact of life. It is the response to error that counts.”
Noninvasive Continuous Positive Airway Pressure (CPAP) Is Dangerous
Mask CPAP is proven to be safe and effective for treating obstructive sleep apnea,2 and noninvasive ventilation has revolutionized the management of acute respiratory failure associated exacerbations of COPD.3 Yet in 1977, the editor of Respiratory Care, Phil Kittredge, published an editorial warning of the grave dangers of using mask CPAP for acute respiratory failure.4
Intermittent Positive-Pressure Breathing for Delivering Aerosol Medications
The idea behind this mode of aerosol delivery is that positive pressure will push medication deeper into the lung where it would be most effective. In truth, intermittent positive-Pressure breathing (IPPB) imparts inertia to aerosol particles increasing oral deposition and decreasing lower airway deposition.5 More importantly, patients with airflow obstruction develop dynamic hyperinflation and occasionally bronchospasm when treated with IPPB.6 Even for those less ill, IPPB is an uncomfortable and time consuming way of taking medication, and so adherence is poor. IPPB has largely been abandoned.
Mist Tent Therapy for Diseases Like Croup and Cystic Fibrosis
The principle that led to this popular therapy was that a cool mist might make it easier to breathe and to expectorate tenacious sputum. However, studies have clearly shown that mist tents have no effect on airway dynamics or outcome in croup.7,8
Moreover, mist tents are difficult to set up and maintain, and deaths can occur in children who become hypoxemic in the mist tent, either when the oxygen supply is inadvertently cut off or when the child's respiratory distress is not easily visualized by caregivers because of the thick fog circulating within the tent. For patients with cystic fibrosis (CF), the high humidity does not help them expectorate secretions or change sputum characteristics9 and after 18 months of mist tent therapy, pulmonary function is worse than those not treated in a mist tent.10
A major concern regarding mist tents is microbial contamination. It is recommended that nebulizer parts be disinfected daily and the plastic tent replaced every day.11 Mold, yeast, and Pseudomonas have been documented growing in the nebulizing chambers of the mist tents used by children with CF.11 Snydman et al12 investigated an in-hospital epidemic of bacteremia in CF patients. This was a very strange epidemic: in most of the patients only the first of 2 blood cultures were positive, and all children “recovered” from their asymptomatic bouts of sepsis, despite only 2 of 11 patients receiving antibiotics. Upon further investigation it was found that the mist tents were contaminated with bacteria and the bacteria was creating pseudo-infections from contamination of the child's skin. Mist tents powered by oxygen also pose a fire hazard, as combustible materials (eg, sheets, blankets, clothing) are maintained in a hyperoxic environment. Despite the lack of efficacy and hazards, mist tents remained popular for pediatric respiratory ailments well into the 1990s and are still being manufactured today.
Normoxia and Eucapnia at All Costs
Until recent times, ventilator strategies used to support patients with ARDS were focused on achieving normoxia and eucapnia; however, large tidal volumes and toxic oxygen concentrations were required to meet these goals. Unfortunately, this approach resulted in more patient deaths, despite normalized blood gasses.13 Even in the 1970s there were concerns about oxygen and ventilator-induced lung injury, but the perceived need to maintain oxygenation and eucapnia outweighed these concerns. Moreover, risk of “barotrauma” was primarily attributed to pneumothoraces from PEEP, not inflation pressure.14
In 1978, Petty and Newman14 recommended that “initially the mechanical ventilator is adjusted with a fairly high tidal volume or 12 to 14 mL per kg.” In an accompanying editorial, Hopewell commented that the available data supported the use of positive-pressure ventilation as a way to preserve surfactant and prevent pulmonary edema.15 However, these cited data also showed that high inflation pressures caused lung injury.16 Animal data strengthened the case that large tidal volumes can cause or worsen lung injury.17 In the 1990s data from human subjects with ARDS emerged suggesting a mortality benefit from avoiding traditionally employed tidal volumes,18,19 culminating in the publication of the landmark ARDS Network trial showing a 22% mortality benefit from low tidal volume ventilation.13
Sedation During Mechanical Ventilation
Agitation, anxiety, and pain are common in patients receiving mechanical ventilation,20 and inadequate sedation may increase the risk of self-extubation.21,22 In response, the practice of inducing deep levels of sedation in patients with continuous infusion of sedatives became commonplace. It was regarded as cruel and dangerous to allow patients to emerge into even light wakefulness. This well intentioned notion turned out to be wrong. The practice of using large doses of sedatives over prolonged periods of time increases the time on the mechanical ventilator, increases the risk of transitioning into delirium, and increases both ICU and hospital stay.23–25
Inhaled N-Acetylcysteine as a Mucolytic
N-acetylcysteine L-cysteine (NAC) has widely been used as a mucolytic to improve secretion clearance. However, aerosolized NAC has never been shown to be effective in COPD26 or CF.27 NAC is rapidly broken down at the airway surface, is acidic, and can cause airway inflammation28 and bronchospasm.29 While oral NAC decreases exhaled hydrogen peroxide (an inflammatory marker),30 nebulized NAC transiently increases exhaled hydrogen peroxide.31
Thus, while there are no data supporting the use of aerosolized NAC for the therapy of any lung disease, and abundant evidence of potential harm, use persists to the extent that a shortage of aerosolized NAC in 2011 had been breathlessly reported in the media as producing panic among physicians and patients.32
Wheezing Is a Sensitive Sign of Airflow Obstruction
Chest auscultation for wheezing has been used as a standard for assessing airflow obstruction and its “improvement” after therapy. There are ample data to suggest that reliance on chest auscultation can lead to under-recognition of obstruction, as well as to the mistaken belief that a therapy has or has not produced benefit.
King et al33 examined 44 adults with normal baseline spirometry who underwent methacholine challenge testing because of a clinical impression of difficult to diagnose asthma. Wheezing was present on maximal forced exhalation in 8 of 14 patients with a positive methacholine challenge test (sensitivity = 57%) and absent in 11 of 30 patients with a negative test (specificity = 37%). However, wheezing on maximal forced exhalation was present in 13 of 27 patients with a PC20 (provocational concentration that produces a 20% FEV1 decrease) greater than 16 mg/mL (no bronchial hyper-responsiveness), and absent in 2 of 7 with a PC20 less than 4 mg/mL.
Chest auscultation for wheezing has also been shown to be unreliable to detect or exclude airflow obstruction in infants34 and older children.35,36 Moreover, judging the effectiveness of bronchodilator therapy on the basis of wheezing intensity after therapy is subjective and correlates very poorly with objective measures of airflow.37 Overreliance on the stethoscope has the potential to result in incorrect clinical decisions.
Dornase Alfa for Diseases Other Than Cystic Fibrosis
Although EBM supports the use of dornase alfa (Pulmozyme, Genentech USA, South San Francisco, California) as a mucolytic for the treatment of CF lung disease,38 there is no proof of efficacy for any other disease, and there is potential for harm, although the risk appears small.39 The unapproved use of dornase alfa for conditions other than CF is expensive, time consuming, of no clinical benefit, and should be discouraged.40
Blow-by Aerosol Administration and Crying During Aerosol Treatments
Aerosol medication deposits best in the airway when patients breathe quietly. There is a misguided perception among some clinicians of the opposite being true, that when children are crying—with short rapid inhalation and prolonged crying exhalation—more medication will get into the lungs. It has also been thought that if you blow the aerosol toward the child's face (“blow-by”), they tolerate it better and will receive equally effective therapy than would be achieved from placing an aerosol mask on their face; this is untrue. Data clearly show that crying substantially reduces aerosol deposition and so-called “blow-by” administration to children with small tidal volumes ensures that almost no medication gets to the lungs. In bench studies, holding the mask 1 cm from the “face” decreases inhaled aerosol by about 60%, and at 2 cm from the “face” the decrease is about 85%, relative to placing the mask directly on the “face.”41–43
Bronchodilators for Tracheomalacia
Patients with severe tracheomalacia often have noisy breathing and wheezing. Because bronchodilators can relax smooth muscle and decrease wheezing in asthma, they have been used to treat the wheezing of tracheomalacia. However, this removes bronchial tone, relaxing the airways so that they are more collapsible, with potentially dire consequences. Paradoxically, a β blocker will increase tracheal smooth muscle tone, thereby improving airway stability and increasing expiratory flows in patients with abnormally collapsible tracheae.44
Cough Medications for Children
Over-the-counter cough and cold medications have been sold for many years, and in 2009 these sales were a $12 billion business in the United States. Despite many studies showing that these medications are ineffective, the United States Food and Drug Administration only urged that these never be used in young children in October of 2007,45 when it was confirmed that these medications were dangerous and had killed children.46–48
The placebo effect for cough therapy is powerful, and the therapeutic benefit of placebo treatment for cough has been reported to be as high as 85%.49 Hutton et al50 found that parents who wanted medicine for their child's cold symptoms at the initial physician visit reported more improvement at follow-up, regardless if the child received drug or placebo.
There is no evidence to support the use of over-the-counter cough medications for the treatment of cough in children or adults; this includes all decongestants, antitussives, expectorants, or mucolytics.51
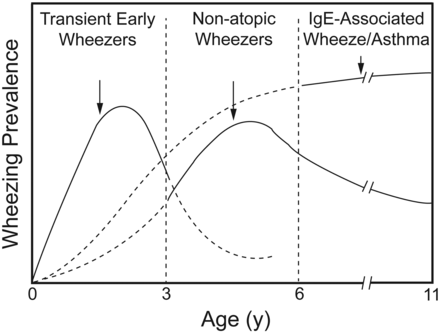
Wheezing in Infants and Young Children Is Probably Asthma
Prospective longitudinal studies show that most children under the age of 3 years who have wheezing do not have asthma, but, rather, are “transient early wheezers” due to virus-induced wheezing (Fig. 1).52–54 In these toddlers with virus-associated wheeze, lung function is often abnormal before the first wheezing episode and even before birth. This has been demonstrated by measuring indices of tidal breathing53 and forced flow at functional residual capacity.55,56 Preschool children with virus-associated wheeze have no evidence of airway eosinophils57 and most have no evidence of asthma. It has been shown that virus-associated wheezing has a good long-term outlook whether treatment is or is not given.58
The Tucson prospective longitudinal study showed that most children under the age of 3 years who wheeze do not have asthma, but, rather, have “transient early wheeze” due to virus. These children obtain no benefit from bronchodilators or corticosteroids, and are not at increased risk of asthma in later life. (From Reference 52, with permission.)
Chronic Cough in Children Is Probably Asthma
Although asthma is a fairly common cause of chronic cough in adults, a large prospective study of chronic cough in children showed that fewer than 5% of these children have asthma.59 Randomized clinical trials have also shown that there is no value in using albuterol for the treatment of chronic cough in children.60 Very few children with chronic cough have “cough variant asthma,” and some pulmonologists are questioning the very existence of this entity.
Exertional Dyspnea in Children Is Probably Asthma
Few children who become short of breath with exercise have asthma or benefit from the use of bronchodilators or inhaled corticosteroids. The cause of exertional dyspnea can usually be determined by formal treadmill exercise testing with physiologic monitoring. A study prospectively evaluating 117 children with exertional dyspnea showed that fewer than 10% had asthma as the cause.61 Asthma appears to be an uncommon cause of cough or dyspnea in children (Table 2).62
Diagnoses of 117 Children After Exercise Testing
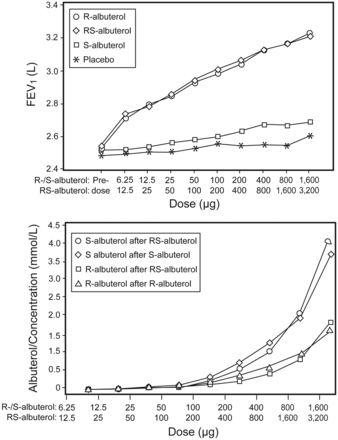
Levalbuterol (Xopenex) Is More Effective and Has Fewer Side Effects Than Racemic Albuterol
Xopenex (R-albuterol, Sunovion Pharmaceuticals [formerly Sepracor], Marlborough, Massachusetts) is the single (dextral) isomer of racemic (RS-) albuterol. Despite promising animal studies suggesting that levalbuterol produces greater airway bronchodilatation, less inflammation, less tremor, and less tachycardia than RS-albuterol, a preponderance of prospective human clinical trials in acute asthma, chronic asthma, life-threatening asthma in children and adults, and COPD have shown that these 2 drugs are identical in efficacy and in side effects (Fig. 2).63–67 The inactive S-albuterol isomer produces no adverse effects (or benefits) in humans. The only clear difference between R-albuterol (Xopenex) and RS-albuterol is that Xopenex is far more expensive.
Levalbuterol (R-albuterol, Xopenex) produces an identical dose-dependent increase in pulmonary function (above) and increase in heart rate (below) as racemic (RS or “usual”) albuterol. The inert isomer in racemic albuterol, S-albuterol, has no effect on either pulmonary function or heart rate. (From Reference 63, with permission.)
Bronchodilator Responsiveness in COPD
COPD is a disease characterized by airflow obstruction in which the FEV1 is reduced in relation to the FVC and the abnormal FEV1 and FEV1/FVC ratio cannot be normalized with medication. While FEV1 improvement can occur in COPD patients treated with bronchodilators, in many patients the FEV1 changes little or not at all. Because of this, some clinicians conclude that these patients do not have bronchodilator responsiveness and would not benefit from bronchodilator therapy. However, bronchodilators can decrease dyspnea in COPD by reducing lung hyperinflation, and the decrease in trapped gas is better measured by changes in thoracic gas volume (ratio of residual volume to total lung capacity), inspiratory capacity, and slow vital capacity. O'Donnell et al68 studied bronchodilator responses in 84 patients with severe emphysema who had less than a 10% increase in FEV1 following bronchodilator. Despite no change in FEV1, most of the patients were found to have significant improvement in lung volume measurements and improved work capacity following bronchodilator administration.
Intravenous Corticosteroids for Exacerbations of Asthma and COPD
Intravenous corticosteroids are often used to treat exacerbations of asthma and COPD, based on the belief that intravenous medications work faster and are more effective. This is not supported by the scientific literature. There was no difference in hospital stay or the ability to decrease the frequency of bronchodilator treatments in children or adults admitted to hospital with asthma who received oral corticosteroids, compared with those who received intravenous steroids.69,70 In subjects with COPD there was no difference in stay, change in pulmonary function, health-related quality of life, or treatment failures in those receiving oral steroids, compared to intravenous therapy.71 There are even data showing that moderate exacerbations of asthma in children and adults can be treated with high dose inhaled corticosteroids.72,73 Intravenous corticosteroids are not only more expensive, but they can potentially take longer to work than oral steroids when the time to insert an intravenous catheter is factored into the equation.
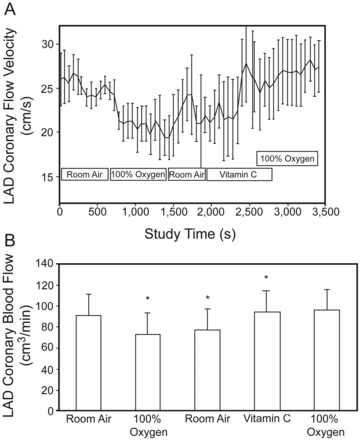
Oxygen Therapy for Chest Pain and Myocardial Infarction
A powerful example of dogma is the strongly held belief that all patients with chest pain due to cardiac ischemia or myocardial infarction (MI) should be treated with supplemental oxygen. Although one might suppose that increasing the blood oxygen content will increase oxygen delivery to the myocardium,73,74 it is documented that in persons who are not hypoxemic, oxygen is a coronary vasoconstrictor (Fig. 3)75,76 and that treatment with 100% oxygen prolongs the duration of electrophysiological abnormalities in patients having an acute MI.77 A randomized trial of oxygen versus air therapy (FIO2 = 0.21) in subjects who had an acute MI showed no difference in mortality, number of arrhythmias, or chest pain.78 A recently published meta-analysis of oxygen therapy in patients with acute MI concluded that the available evidence shows no benefit in using oxygen to treat patients with acute MI, and there was a suggestion of increased mortality risk associated with oxygen therapy.79
The effect of 100% oxygen administration on left anterior descending (LAD) coronary artery blood flow and velocity in patients with ischemic heart disease. (From Reference 75, with permission.)
The Use of “Percent of Predicted” to Classify Pulmonary Function Data
The use of 80% of the predicted mean value to define the lower limit of normal pulmonary function data is a widespread practice. However, the use of “percent of predicted” to define the limits of normality often misclassifies patients (Fig. 4).80,81 In 1979, Sobol and Sobol81 wrote that “nowhere else in medicine is such a naïve view taken of the lower limit of normal.” If the 80% of predicted rule were correct, the lower limit of normal for arterial oxyhemoglobin saturation would be 76% (95% as the mean of normality). In a similar way, the use of a fixed FEV1/FVC ratio (< 70%) to diagnose airflow obstruction can produce errors in COPD diagnosis, since a ratio < 70% is often a normal finding in the aged lung.82 The lower and upper limits of normal can better be statistically defined as the fifth percentile, rather than a percent of predicted.
Percentage of subjects of different ages (n = 11,413 pulmonary function tests) who would be misclassified if a fixed ratio of FEV1/FVC of ≤ 70% was used to define airflow obstruction. (From Reference 80, with permission.)
Evidence-Based Medicine: Basic Elements of Good Research
Study Design
The first step in conducting a research study is to ask the right questions. A question is important and novel when it can change our understanding—like a new idea or a new way of looking at an old idea or question. The question should be interesting and detailed enough so that the investigator can specify a testable hypothesis. Furthermore, there should be preliminary data to support the hypothesis being tested. Ask irrelevant questions, and you're likely to get irrelevant answers. For example, in the 1930s a statistical exercise found nearly perfect correlation between the stork and human populations in Oldenburg, Germany (Fig. 5). One author commented, “anyone who draws the incorrect conclusion that storks bring babies and proceeds to shoot storks in the hopes of reducing the population will be disappointed!”83,84
Correlation between the end-of-year human and stork populations from years 1930–1936 in Oldenburg, Germany. (From Reference 83, with permission).
The hypothesis and specific aims must be clearly stated before beginning a study. It is important to state what results you hypothesize will be shown (so that the hypothesis can potentially be rejected), demonstrate that the hypothesis is plausible, and specify how you will accept or reject the hypothesis using the primary outcome variable(s) identified before the study begins. Outcome measures used for answering the research question need to be relevant, scientifically validated, and testable. It is important to involve a statistician during the study design, and not engage a statistician only after data collection is complete, for the purposes of data analysis.
The methods for conducting a study need to be completely and accurately detailed, so that anyone reading the study can reproduce your methods. The study should be appropriately powered, well conducted, analyzed accurately, and finally it must be reported in the peer-reviewed literature. Power calculations are critically important and are a common problem with study design. There are readily available on-line programs that can help with calculating statistical power; for example, the free software G*Power (link: http://www.psycho.uni-duesseldorf.de/abteilungen/aap/gpower3/).
An underpowered study can lead to a Type 2 error, or failure to find statistically significant differences because of an inadequate sample size. In many ways it is just as bad to have a study that is over-powered. If a study is over-powered, in other words, more subjects are studied than necessary, the duration of the study is lengthened, the cost of the study increases dramatically, patients may remain on a less effective therapy for a longer period of time awaiting the completion of the study, and there is a risk of documenting a statistically significant but not a clinically relevant difference between therapies studied.
It is important to identify an appropriate control group for the study, taking into account complicating factors such as concomitant medical conditions, other medications taken, disease severity, time of the year, and adherence when taking medications. There should be a mix of sex, race, and age if these are thought to influence the disease in question. Randomization also needs to be done in advance, using acceptable randomization techniques. The purpose of randomization is to distribute confounding factors equally between treatment groups. Although randomization may not be needed for crossover studies, a crossover design will increase the duration of the study, and there is always the possibility of carry-over effects confounding data interpretation.
Regardless of the population studied, documentation of informed consent and approval of the study by an institutional review board (IRB) before starting the study are requirements for publication, and all clinical trials conducted in the United States should be registered with the National Institutes of Health (http://www.clinicaltrials.gov) before enrolling the first subject. Informed consent can be difficult or impossible for patients who may not be fully alert and have the ability to be well informed, such as those in the emergency department or an ICU, and children. While it is important to do studies in these populations, an ethicist should be involved to ensure the safety of these vulnerable patients. There is also the issue of control subjects potentially having effective therapy withheld. This needs to be accounted for so that subjects in the placebo arm are not placed at risk. Nevertheless, controls are essential, because just being enrolled in a study can change adherence, expectations, and outcomes. This is sometimes called the Hawthorne effect.
Interpreting and Reporting the Data
All aspects of clinical trials need to be well documented, including initial questions and hypotheses, methods (statistical and non-statistical), IRB and ethics approval, how subjects were recruited, and how many patients elected not to participate in the study or dropped out after entering the study. When considering the application of the findings from a clinical trial to bedside care, clinicians must keep in mind that clinical trials are conducted on select individuals, while the clinician treats all patients who present with a particular problem. For example, Brochard et al3 reported reduced stay, need for intubation, and mortality in subjects with COPD exacerbations treated with noninvasive ventilation. The investigators screened 275 subjects, but only 85 entered the study. While clinical trials need to have specific inclusion criteria, it is important to ask if the results of any study would have been the same if all or most of the screened patients entered the trial. In the Brochard study, 17% of screened subjects were excluded because of concomitant heart failure, 9% had pneumonia or sepsis, 6% had asthma, and 34% were excluded for “various reasons.” Do the results of the Brochard study apply to all or most COPD exacerbation patients? Should the clinician expect the same results from a clinical trial when treating all comers at the bedside? Brochard et al3 reported an intubation rate of 26% in the treatment arm of the study. Would the intubation rate have been higher if the study protocol was applied to COPD patients with concomitant heart failure, sepsis, pneumonia, and asthma? Perhaps, but again, perhaps not.
Subjects who are lost to follow-up may differ in important ways from those who remain in the study until completion. Subjects may get well and leave the study, or may become worse or die. Moreover, subjects experiencing more side effects may choose to drop out from the study protocol. Therefore, every subject enrolled in a study should be accounted for, and an intent-to-treat data analysis, as well as a fully adherent data analysis, needs to be conducted.85 Intent-to-treat analysis preserves the effect of randomization and gives a better estimate of the real life impact of a study. For example, suppose there was a study conducted comparing an aggressive surgical intervention against more conservative medical management of an acute illness. The primary outcome for the study is 28-day survival. Those patients arriving at the emergency department who were randomized to the medical group had therapy initiated immediately, while those randomized to surgery were transferred to the surgical suite where they were prepped for surgery. At the end of the trial it is reported that the patients who underwent surgery had somewhat better outcomes than those treated more conservatively. However, suppose the researchers excluded a substantial number of patients from the surgical arm of the study because they died while waiting for surgery. The researchers chose not to include these patients because they didn't receive the surgical intervention; however, in real life these patients still would have died! By using intent-to-treat analysis, the patients who died preoperatively would be included in the survival comparison, and the reported study findings might be quite different.
The peer review process is a critical component of EBM. It is the right of a journal editor or reviewer to request and review all study documentation if there are any questions. Moreover, journal editors consult with expert statisticians if there are questions regarding the statistical methods used to collect, analyze, and report data.
It is required to document and report all adverse events, even if these events are not believed to be treatment-related. Sometimes there are safety or efficacy evaluations done by an independent committee in order to avoid placing subjects at unnecessary risks. Follow-up after the study should be completed for as many patients as possible. Secondary data analysis is useful to ask questions for subsequent studies, but should never be reported as a major finding, even if the results are statistically significant.
It is important to state precisely how large a treatment effect was observed. This is often done by reporting a 95% CI, which denotes a 1-in-20 chance of a Type 1 error. The difference between clinical relevance and statistical significance is extremely important to consider, especially when evaluating the meaning of mega-studies enrolling tens of thousands of subjects. To illustrate the difference, suppose that there is a new therapy being studied that prolongs survival of one form of lung cancer. A large clinical trial of thousands of subjects randomized to receive usual care plus this new therapy or plus placebo shows a significantly increased survival time in those receiving the new therapy (P < .05). Although this is statistically significant, if the increased survival was 50 weeks, compared to 49 weeks for those on placebo, the drug cost $50,000 per year, and there were a large number of side effects with the drug, this difference would not be clinically important. Indeed, when other factors are taken into account, such a therapy could well be considered to be detrimental. Another statistical estimate of the impact of a therapy is the number-needed-to-treat (NNT). The NNT estimates how many patients need to be exposed to a therapy for one patient to avoid a bad outcome. Akobeng86 provided a hypothetical example of how a statistically significant finding can be put into clinical context with NNT analysis. In this example, a fictional drug reduces 5-year mortality risk by 80%; however, NNT analysis tempers this impressive statistic by revealing that 25,000 patients would have to take the medication for 5 years to prevent one death.
Limitations of Evidence-Based Medicine
EBM is not panacea. Grading of evidence can be subjective, and there can be publication bias because negative studies are often not published. This may be due to “negative” results resulting from a Type 2 error (underpowered study due to insufficient subjects studied to reach statistical significance), but it could also be due to there being intrinsically less interest in negative studies. Meta-analysis may be important for pooling small studies; the best-known example is the Cochrane Collaboration, found at http://www.cochrane.org. Even a “good” study design can be influenced by investigator bias or by a sponsor who funds the study. This is extremely important information to know when trying to generalize study results to a broader patient population. Finally, remember that EBM is not “cook book” medicine, and not all well conducted studies are necessarily well designed, interesting, important, or even meaningful. We live in the era of EBM; however, evidence is of limited value if not applied consistently.87,88
Conclusions
There have been many examples in respiratory care of dogma perpetuating poor care for our patients, even in the face of clear evidence demonstrating ineffectiveness or harm. It is our duty as professionals to question dogma, to apply evidence to all of our professional duties, and when evidence is not available, to do the well designed studies that will lead to answers.
Footnotes
- Correspondence: Bruce K Rubin MD MEngr MBA FAARC, Children's Hospital of Richmond at Virginia Commonwealth University, Richmond VA 23298. E-mail: brubin{at}vcu.edu.
Dr Rubin presented a version of this paper as the 27th Philip Kittredge Memorial Lecture at the AARC Congress 2011, held November 5–8, 2011, in Tampa, Florida.
- Copyright © 2012 by Daedalus Enterprises Inc.
References
- 1.↵
- 2.↵
- 3.↵
- 4.↵
- 5.↵
- 6.↵
- 7.↵
- 8.↵
- 9.↵
- 10.↵
- 11.↵
- 12.↵
- 13.↵
- 14.↵
- 15.↵
- 16.↵
- 17.↵
- 18.↵
- 19.↵
- 20.↵
- 21.↵
- 22.↵
- 23.↵
- 24.
- 25.↵
- 26.↵
- 27.↵
- 28.↵
- 29.↵
- 30.↵
- 31.↵
- 32.↵
- 33.↵
- 34.↵
- 35.↵
- 36.↵
- 37.↵
- 38.↵
- 39.↵
- 40.↵
- 41.↵
- 42.
- 43.↵
- 44.↵
- 45.↵
- 46.↵
- 47.
- 48.↵
- 49.↵
- 50.↵
- 51.↵
- 52.↵
- 53.↵
- 54.↵
- 55.↵
- 56.↵
- 57.↵
- 58.↵
- 59.↵
- 60.↵
- 61.↵
- 62.↵
- 63.↵
- 64.
- 65.
- 66.
- 67.↵
- 68.↵
- 69.↵
- 70.↵
- 71.↵
- 72.↵
- 73.↵
- 74.↵
- 75.↵
- 76.↵
- 77.↵
- 78.↵
- 79.↵
- 80.↵
- 81.↵
- 82.↵
- 83.↵
- 84.↵
- 85.↵
- 86.↵
- 87.↵
- 88.↵