Abstract
Purpose
Pneumonia associated with invasive mechanical ventilation (IMV) is one of the indicator infections of the German Nosocomial Infection Surveillance System. In 2005 surveillance was extended to include pneumonia associated with noninvasive ventilation (NIV). The aim of this study was to determine the utilization of IMV and NIV and the associated incidence densities (IDs) of pneumonia and to compare the characteristics of pneumonia cases and the spectrum of associated pathogens.
Methods
We analyzed the pooled data of 400 intensive care units (ICUs) with respect to three categories of pneumonia: pneumonia associated with IMV and NIV and pneumonia not associated with ventilation. Pooled ventilation utilization rates and pneumonia IDs were calculated in total and stratified by hospital size, hospital type and ICU type.
Results
Four hundred ICUs with 779,500 admitted patients, 1,068,472 IMV days and 101,569 NIV days reported 6,869 cases of pneumonia between 2005 and 2007. Of these, 5,811 cases were associated with IMV, 160 with NIV and 898 were not associated with ventilation. The mean pneumonia IDs were 1.58 and 5.44 cases per 1,000 ventilator days for NIV and IMV, respectively. Pneumonia cases associated with IMV were younger, had a longer ICU stay before onset of pneumonia and were more often associated with gram-negative bacteria than cases associated with NIV; however, there were no differences in the proportion of secondary sepsis and death.
Conclusions
This surveillance study including pneumonia associated with IMV and NIV and pneumonia not associated with ventilation shows significant differences of pneumonia IDs, patient characteristics and the spectrum of associated pathogens.
Similar content being viewed by others
Introduction
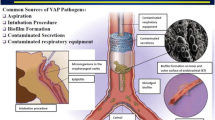
Ventilator-associated pneumonia (VAP) is a serious and common complication of intensive care unit (ICU) treatment and is associated with a longer ICU stay, higher morbidity and mortality, increased consumption of resources and additional hospital costs [1, 2]. VAP rates can be reduced through multiple interventions such as for instance education programmes and VAP prevention bundles as well as through surveillance [3–5]. VAP is included as one of the indicator infections in nosocomial infection surveillance systems [6, 7].
Noninvasive ventilation (NIV) is used for an increasing range of indications [8–14], has been associated with lower rates of serious complications, especially of nosocomial pneumonia [15–19], and the success of NIV has been found to be associated with improved survival [20]. NIV has been described among other measures as a potential strategy to prevent VAP [21] and the recommendation to use NIV whenever possible has been incorporated in guidelines for VAP prevention [22, 23].
The ICU module of the German Nosocomial Infection Surveillance System (ICU-KISS) has collected data on pneumonia associated with invasive mechanical ventilation (IMV) in individual ICUs and provided national reference rates since 1997. In 2005, surveillance was extended to include pneumonia associated with noninvasive ventilation in addition to invasive mechanical ventilation. The aim of this study was to determine the utilization of invasive and noninvasive ventilation and the associated incidence densities (IDs) of pneumonia in 400 German ICUs participating in the ICU-KISS module between 2005 and 2007 and to compare characteristics of pneumonia cases associated with different types of ventilation and the spectrum of associated pathogens.
Methods
Detailed methods of ICU-KISS have been described previously [5, 7, 24, 25]. In short, ICU-KISS uses a unit-based approach and determines the number of patients, patient days and device days of individual ICUs as a denominator for the calculation of device-associated infection rates. Only in case of occurrence of nosocomial infections are patient-based data collected. Ventilator-associated pneumonia is one of the three indicator infections besides central venous catheter-associated bloodstream infection and urinary catheter-associated urinary tract infection used to evaluate infection control management in ICUs. Data are collected by infection control personnel in individual ICUs and is submitted via a web-based surveillance portal to the central database of the national reference centre. Participants can use the data analysis function of the surveillance portal at any time to compare local IDs of indicator infections with the national reference calculated from the pooled data of all participating ICUs.
To enable comparisons between ICUs, standardized definitions for devices and nosocomial infections are used. Invasive mechanical ventilation is defined as continuous ventilation via endotracheal tube or tracheostomy and noninvasive ventilation as positive pressure ventilation with different pressure levels via face mask or helmet performed intermittently or continuously for at least 6 h per day. The use of continuous positive airway pressure (CPAP) alone is not regarded as noninvasive ventilation.
Nosocomial pneumonia is diagnosed according to CDC definitions from a combination of clinical, laboratory and radiological criteria as “clinically defined pneumonia” or with additional microbiologic results on associated pathogens as “pneumonia with common bacterial or fungal pathogens” or “atypical pneumonia” [6]. There are additional pneumonia definitions for pneumonia in immunocompromised patients and for infants and children less than 12 years old.
Pneumonia is considered to be associated with NIV or IMV if ventilation has been performed for at least 6 h (NIV) or at least 12 h (IMV) during the 48-h period before onset of pneumonia. If NIV as well as IMV has been performed within 48 h of the onset of pneumonia, pneumonia is considered to be associated with IMV. Pneumonia is considered to be not associated with ventilation if neither IMV nor NIV has been performed within 48 h before the onset of pneumonia for the minimum period described above.
For each episode of nosocomial pneumonia the following data are collected: date of ICU admission and of onset of pneumonia, age and sex of the patient, up to four associated pathogens and the material from which pathogens were isolated (tracheal secretion, bronchoalveolar lavage/protected specimen brush, blood culture, other) and the occurrence of complications such as secondary sepsis and death during the ICU stay.
Pooled mean and median ventilation utilization rates per 100 patient days with 25 and 75% quantiles and mean and median incidence densities (IDs) of pneumonia with 25 and 75% quantiles per 1,000 ventilation days were calculated. The following pneumonia IDs were calculated: pneumonia per 1,000 patient days without ventilation for pneumonia not associated with ventilation, pneumonia associated with IMV per 1,000 IMV days and pneumonia associated with NIV per 1,000 NIV days. Differences between ventilation utilization and associated pneumonia IDs were tested by the Wilcoxon test for two categories and the Kruskal-Wallis test for more than two categories or the chi-square test. The significance level was P < 0.05. All analyses were performed with SPSS version 14.0.
Results
A total of 474 ICUs participated in ICU-KISS between 2005 and 2007. Of these, 400 (84.4%) performed NIV, and 74 (15.6%) did not document any NIV days during the 3-year observation period. ICUs performing NIV did not differ from ICUs that did not perform NIV with respect to hospital type (university hospital, academic hospital, other hospital), hospital size (<600 beds vs. ≥600 beds) and ICU type (medical, surgical, medical-surgical). Further analysis was based on data of the 400 ICUs performing IMV and NIV, and the characteristics of these ICUs are shown in Table 1.
Pooled mean ventilation utilization rates were 3.75 per 100 patient days for NIV and 39.42 per 100 patient days for IMV and pooled mean ventilator-associated pneumonia IDs were 1.58 and 5.44 cases per 1,000 ventilation days for NIV and IMV, respectively. The mean ID of pneumonia not associated with ventilation was lower with 0.58 cases per 1,000 patient days without ventilation.
For IMV but not for NIV there were significant differences in the utilization of ventilation between ICUs in small and large hospitals as well as between ICUs in university, teaching and other hospitals and ICUs of different types (Table 2). The hospital type and size did not influence pneumonia IDs associated with IMV, only the type of ICU affected pneumonia IDs with surgical ICUs having significantly higher median pneumonia IDs than other types of ICUs (Table 2). No significant differences of pneumonia IDs could be determined in any of the categories (hospital type and size, ICU type) for pneumonia not associated with ventilation and pneumonia associated with NIV.
A total of 6,869 cases of nosocomial pneumonia occurred during the analyzed 3-year period. Of these, 5,811 cases were associated with IMV, 160 were associated with NIV, and 898 were not associated with ventilation. The patient characteristics of these pneumonia cases are shown in Table 3. Cases of the three categories of pneumonia differed significantly by age, duration of ICU stay before onset of pneumonia, and the proportion of secondary sepsis and death. Patients with pneumonia associated with IMV were younger and had a longer ICU stay before onset of pneumonia than patients with pneumonia associated with NIV or patients without ventilation. Pneumonia cases associated with ventilation (IMV and NIV) had a higher proportion of secondary sepsis and death in comparison to pneumonia cases without ventilation, but there was no difference in this respect between the two modes of ventilation.
The proportion of cases with clinically defined pneumonia and with pneumonia with bacterial confirmation differed significantly between the three pneumonia categories (P < 0.001). Pathogens could be recovered in more patients with pneumonia undergoing IMV than in patients without ventilation or in patients receiving NIV. The numbers of cases of pneumonia associated with selected pathogens and their respective proportions per 100 pathogens are listed in Table 4. Pathogens are not listed by pneumonia cases, because due to the higher proportion of cases with isolated pathogens in the IMV pneumonia category, there was also a significantly higher proportion of nearly all specific pathogens in pneumonia associated with IMV.
Significant differences in the spectrum of associated pathogens per 100 pathogens were found for streptococci (higher proportion in pneumonia associated with NIV and in pneumonia not associated with ventilation), all gram-negative bacteria (higher proportion in ventilated patients) and Pseudomonas aeruginosa (higher proportion in ventilated patients). There were also significant differences (P < 0.001) in the duration from ICU admission to pneumonia for cases associated with different pathogens (data not shown). Especially pneumonia cases associated with P. aeruginosa showed a longer interval between ICU admission and pneumonia with a median duration of 12 days (NIV group) and 13 days (IMV group) in cases of pneumonia associated with P. aeruginosa compared to 9 days (NIV group) and 10 days (IMV group) in cases of pneumonia associated with other pathogens (P = 0.036).
Discussion
In this study we evaluated the incidence and the characteristics of three categories of nosocomial pneumonia—pneumonia associated with invasive mechanical ventilation, pneumonia associated with noninvasive mechanical ventilation and pneumonia not associated with ventilation—using data collected during continuous active surveillance in 400 German ICUs participating in the German nosocomial infection surveillance system.
Pneumonia associated with NIV has been difficult to characterize because it is a rare event and studies investigating pneumonia associated with NIV often include only few cases. A recent review that pooled data on pneumonia associated with NIV from 12 studies included a total of 38 cases with less than 10 pneumonia cases reported in 11 of the 12 studies [18]. In this study we were able to document 160 cases of NIV-associated pneumonia using homogenous definitions, methods and criteria with the pooled data of 400 ICUs from a 3-year period. The low incidence of NIV-associated pneumonia is illustrated in our study by the fact that more than half of the participating ICUs did not document a single case of NIV-associated pneumonia during the analyzed period resulting in a median incidence density of 0 cases per 1,000 NIV days.
Our analysis shows that patient characteristics such as age and length of stay differed significantly between the analyzed categories of pneumonia cases. The finding that different patient populations might be affected by IMV- and NIV-associated pneumonia should be taken into account in the interpretation of studies regarding the benefit of NIV for prevention of pneumonia. Although patients receiving NIV in our study had the advantage of a reduced incidence of pneumonia, at the time they developed pneumonia the proportion of secondary sepsis and death was as high as in patients with IMV-associated pneumonia.
Pathogens were significantly more often recovered in patients with pneumonia associated with IMV, presumably due to the easier availability of material for culture, while more cases of pneumonia associated with NIV and pneumonia not associated with ventilation were diagnosed based on clinical findings only. The pathogens most often recovered in this study—Staphylococcus aureus and Pseudomonas aeruginosa—were the same as the ones most often reported to the NHSN system and in two large epidemiologic pneumonia studies [26–28]; however, starting from the third position, the composition of pathogens differs between the studies. Unfortunately, pathogens associated with pneumonia cases in our study cannot always be interpreted as causative or the only causative pathogens, because up to four pathogens could be reported per pneumonia episode. Especially Candida spp. are unlikely to play a large role as the cause of pneumonia.
The analysis of isolated pathogens showed significant differences in the spectrum of pathogens associated with the three pneumonia categories. However, it remains unclear whether these differences are directly related to the mode of ventilation or whether they are caused by differences regarding the patient population or the duration between ICU admission and onset of pneumonia in the three pneumonia categories. Especially the duration of ventilation prior to onset of pneumonia is known to influence the spectrum of isolated pathogens [29], and in our study there was significant variation in the interval between ICU admission and onset of pneumonia in the three groups. The differences in the spectrum of pathogens with for example a lower proportion of Streptococci and a higher proportion of gram-negative bacteria in the IMV group who has the longest interval between admission and pneumonia are similar to the differences that have been described for early onset and late-onset pneumonia [23].
The reduced incidence of ventilator-associated pneumonia in patients receiving NIV in comparison to IMV has been attributed to the avoidance of endotracheal intubation. Endo-tracheal tubes have been shown to provide easy access for bacteria to the lower respiratory tract by impairing the mucociliary clearance and the cough reflex, providing a surface for biofilm formation and by facilitating microaspirations from the colonized oropharyngeal tract [30]. It has even been suggested that the term “endotracheal intubation related pneumonia” would be more appropriate than “ventilator-associated pneumonia” [31], and multiple prevention measures are focused on reducing the adverse effects of the endotracheal tube [32]. While in our study the mean pneumonia ID in patients receiving IMV was four times higher than for patients receiving NIV, our data also show that NIV is associated with a threefold increase of the pneumonia ID in comparison to no ventilation, suggesting that ventilation is associated with a higher risk for pneumonia also in the absence of the endotracheal tube.
A limitation of this study is that ICU-KISS does not collect data to describe the ICU patient population with regard to underlying diseases, disease severity or other known risk factors for nosocomial pneumonia, because it uses a unit-based and not a patient-based surveillance approach. For the same reason ICU-KISS does not collect data on the indication for and the duration of ventilation in individual patients. Structural data of ICUs can been used as a surrogate marker for different patient populations with for instance ICUs in larger or university hospitals in general caring for patients with higher disease severity. Contrary to this assumption, the analysis of structural data showed that only the ICU type and not the hospital size or the hospital type affected pneumonia IDs in patients receiving IMV. Surgical ICUs had significantly higher pneumonia IDs than the other ICUs confirming that surgical patients are a high-risk group for ventilator-associated pneumonia and should be targeted preferentially by prevention measures. This is in accordance with the finding that surgery itself—emergency surgery and elective surgery—is an independent risk factor for nosocomial pneumonia [33].
The use of NIV compared to IMV in this sample of ICUs seems to be low; however, the extent of documented NIV days might have been influenced by the strict criteria of at least 6 h of daily NIV use. Although it has been shown that NIV use has increased in response to published clinical research [34], 74 ICUs in our study did not document any NIV days, indicating that NIV has not been integrated systematically into practice in all ICUs. Patients in these ICUs might benefit from the education of clinicians about noninvasive ventilatory strategies as recommended in guidelines [22].
In conclusion, this surveillance study demonstrates that surveillance of pneumonia associated with noninvasive ventilation is feasible in a large sample of 400 ICUs. Comparison of pneumonia associated with noninvasive ventilation to pneumonia associated with invasive ventilation and pneumonia not associated with ventilation shows significant differences of pneumonia incidence densities, patient characteristics and the spectrum of associated pathogens.
References
Safdar N, Dezfulian C, Collard HR, Saint S (2005) Clinical and economic consequences of ventilator-associated pneumonia: a systematic review. Crit Care Med 33:2184–2193
Hugonnet S, Eggimann P, Borst F, Maricot P, Chevrolet JC, Pittet D (2004) Impact of ventilator-associated pneumonia on resource utilization and patient outcome. Infect Control Hosp Epidemiol 25:1090–1096
Salahuddin N, Zafar A, Sukhyani L, Rahim S, Noor MF, Hussain K, Siddiqui S, Islam M, Husain SJ (2004) Reducing ventilator-associated pneumonia rates through a staff education programme. J Hosp Infect 57:223–227
Hawe CS, Ellis KS, Cairns CJ, Longmate A (2009) Reduction of ventilator-associated pneumonia: active versus passive guideline implementation. Intensive Care Med 35:1180–1186
Zuschneid I, Schwab F, Geffers C, Behnke M, Rüden H, Gastmeier P (2007) Trends in ventilator-associated pneumonia rates within the German nosocomial infection surveillance system (KISS). Infect Control Hosp Epidemiol 28:314–318
Horan TC, Andrus M, Dudeck MA (2008) CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 36:309–332
Gastmeier P, Sohr D, Schwab F, Behnke M, Zuschneid I, Brandt C, Dettenkofer M, Chaberny IF, Rüden H, Geffers C (2008) Ten years of KISS: the most important requirements for success. J Hosp Infect 70(Suppl.1):11–16
Antonelli M, Conti G, Rocco M, Bufi M, De Blasi RA, Vivino G, Gasparetto A, Meduri GU (1998) A comparison of noninvasive positive-pressure ventilation and conventional mechanical ventilation in patients with acute respiratory failure. N Engl J Med 339:429–435
Antonelli M, Conti G, Bufi M, Costa MG, Lappa A, Rocco M, Gasparetto A, Meduri GU (2000) Noninvasive ventilation for treatment of acute respiratory failure in patients undergoing solid organ transplantation: a randomized trial. JAMA 283:235–241
Hilbert G, Gruson D, Vargas F, Valentino R, Gbikpi-Benissan G, Dupon M, Reiffers J, Cardinaud JP (2001) Noninvasive ventilation in immunosuppressed patients with pulmonary infiltrates, fever, and acute respiratory failure. N Engl J Med 344:481–487
Rocco M, Conti G, Antonelli M, Bufi M, Costa MG, Alampi D, Ruberto F, Stazi GV, Pietropaoli P (2001) Non-invasive pressure support ventilation in patients with acute respiratory failure after bilateral lung transplantation. Intensive Care Med 27:1622–1626
Azoulay E, Alberti C, Bornstain C, Leleu G, Moreau D, Recher C, Chevret S, Le Gall JR, Brochard L, Schlemmer B (2001) Improved survival in cancer patients requiring mechanical ventilatory support: impact of noninvasive mechanical ventilatory support. Crit Care Med 29:519–525
Lefebvre A, Lorut C, Alifano M, Dermine H, Roche N, Gauzit R, Regnard JF, Huchon G, Rabbat A (2009) Noninvasive ventilation for acute respiratory failure after lung resection: an observational study. Intensive Care Med 35:663–670
Benditt JO (2009) Novel uses of noninvasive ventilation. Respir Care 54:212–219
Guérin C, Girard R, Chemorin C, De Varax R, Fournier G (1997) Facial mask noninvasive mechanical ventilation reduces the incidence of nosocomial pneumonia: a prospective epidemiological survey from a single ICU. Intensive Care Med 23:1024–1032
Nourdine K, Combes P, Carton MJ, Beuret P, Cannamela A, Ducreux JC (1999) Does noninvasive ventilation reduce the ICU nosocomial infection risk? A prospective clinical survey. Intensive Care Med 25:567–573
Girou E, Schortgen F, Delclaux C, Brun-Buisson C, Blot F, Lefort Y, Lemaire F, Brochard L (2000) Association of noninvasive ventilation with nosocomial infections and survival in critically ill patients. JAMA 284:2361–2367
Hess DR (2005) Noninvasive positive-pressure ventilation and ventilator-associated pneumonia. Respir Care 50:924–929
Girou E, Brun-Buisson C, Taillé S, Lemaire F, Brochard L (2003) Secular trends in nosocomial infections and mortality associated with noninvasive ventilation in patients with exacerbation of COPD and pulmonary edema. JAMA 290:2985–2991
Demoule A, Girou E, Richard JC, Taille S, Brochard L (2006) Benefits and risks of success or failure of noninvasive ventilation. Intensive Care Med 32:1756–1765
Koenig SM, Truwit JD (2006) Ventilator-associated pneumonia: diagnosis, treatment, and prevention. Clin Microbiol Rev 19:637–657
Coffin SE, Klompas M, Classen D, Arias KM, Podgorny K, Anderson DJ, Burstin H, Calfee DP, Dubberke ER, Fraser V, Gerding DN, Griffin FA, Gross P, Kaye KS, Lo E, Marschall J, Mermel LA, Nicolle L, Pegues DA, Perl TM, Saint S, Salgado CD, Weinstein RA, Wise R, Yokoe DS (2008) Strategies to prevent ventilator-associated pneumonia in acute care hospitals. Infect Control Hosp Epidemiol 29(Suppl 1):31–40
American Thoracic Society; Infectious Diseases Society of America (2005) Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 171:388–416
Gastmeier P, Geffers C, Sohr D, Dettenkofer M, Daschner F, Rüden H (2003) Five years working with the German nosocomial infection surveillance system (Krankenhaus Infektions Surveillance System). Am J Infect Control 31:316–321
Meyer E, Sohr D, Gastmeier P, Geffers C (2009) New identification of outliers and ventilator-associated pneumonia rates from 2005 to 2007 within the German Nosocomial Infection Surveillance System. J Hosp Infect 73:246–252
Hidron AI, Edwards JR, Patel J, Horan TC, Sievert DM, Pollock DA, Fridkin SK, National Healthcare Safety Network Team; Participating National Healthcare Safety Network Facilities (2008) NHSN annual update: antimicrobial-resistant pathogens associated with healthcare-associated infections: annual summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2006–2007. Infect Control Hosp Epidemiol 29:996–1011
Kollef MH, Shorr A, Tabak YP, Gupta V, Liu LZ, Johannes RS (2005) Epidemiology and outcomes of health-care-associated pneumonia: results from a large US database of culture-positive pneumonia. Chest 128:3854–3862
Koulenti D, Lisboa T, Brun-Buisson C, Krueger W, Macor A, Sole-Violan J, Diaz E, Topeli A, DeWaele J, Carneiro A, Martin-Loeches I, Armaganides A, Rello J, EU-VAP/CAP Study Group (2009) Spectrum of practice in the diagnosis of nosocomial pneumonia in patients requiring mechanical ventilation in European intensive care units. Crit Care Med 37:2360–2368
Park DR (2005) The microbiology of ventilator-associated pneumonia. Respir Care 50:742–763
Pneumatikos IA, Dragoumanis CK, Bouros DE (2009) Ventilator-associated pneumonia or endotracheal tube-associated pneumonia? An approach to the pathogenesis and preventive strategies emphasizing the importance of endotracheal tube. Anesthesiology 110:673–680
Safdar N, Crnich CJ, Maki DG (2005) The pathogenesis of ventilator-associated pneumonia: its relevance to developing effective strategies for prevention. Respir Care 50:725–739
Ramirez P, Ferrer M, Torres A (2007) Prevention measures for ventilator-associated pneumonia: a new focus on the endotracheal tube. Curr Opin Infect Dis 20:190–197
Wolkewitz M, Vonberg RP, Grundmann H, Beyersmann J, Gastmeier P, Bärwolff S, Geffers C, Behnke M, Rüden H, Schumacher M (2008) Risk factors for the development of nosocomial pneumonia and mortality on intensive care units: application of competing risk models. Crit Care 12:R44
Esteban A, Ferguson ND, Meade MO, Frutos-Vivar F, Apezteguia C, Brochard L, Raymondos K, Nin N, Hurtado J, Tomicic V, González M, Elizalde J, Nightingale P, Abroug F, Pelosi P, Arabi Y, Moreno R, Jibaja M, D’Empaire G, Sandi F, Matamis D, Montanez AM, Anzueto A, VENTILA group (2008) Evolution of mechanical ventilation in response to clinical research. Am J Respir Crit Care Med 177:170–177
Acknowledgments
The authors wish to thank the health care staff and the infection control personnel of all KISS-ICUs for their participation and the support of this surveillance project.
Conflict of interest statement
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kohlenberg, A., Schwab, F., Behnke, M. et al. Pneumonia associated with invasive and noninvasive ventilation: an analysis of the German nosocomial infection surveillance system database. Intensive Care Med 36, 971–978 (2010). https://doi.org/10.1007/s00134-010-1863-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-010-1863-z