Abstract
Purpose
Lung tissue may exhibit a biochemical response to excessive deformation. Since strain has been proposed as a marker of such deformation, we studied the relationships between strain and matrix remodeling and inflammation markers in mechanically ventilated patients with and without acute lung injury (ALI).
Methods
Twenty-two ventilated patients were studied (16 with ALI, 6 controls). Clinical data, gas exchange and respiratory mechanics were recorded, and end-expiratory lung volume (EELV) was measured by oxygen washin/washout. Extracellular matrix remodeling markers (procollagen, matrix metalloproteinases -2 and -9, TIMP-1) and inflammation markers (IL-6, IL-8, IL-10, IFNγ, IL-17A, and VEGF) were measured in bronchoalveolar lavage fluid (BALF). Strain was computed as the ratio between tidal volume and EELV. Patients with ALI were divided into two subgroups according to the median strain value (0.27).
Results
Patients in the ALI group exhibited higher airway pressures, lower EELV and higher strain than the control group. There were no significant differences in gas exchange, respiratory mechanics, or the matrix remodeling markers between ALI patients with normal and high strain. The subgroup of patients with high strain showed a fourfold increase of IL-6 and IL-8 concentrations in BALF, compared with patients with ALI and normal strain or patients without ALI. In the whole sample, IL-6 and IL-8 concentrations in BALF were correlated with strain (Spearman’s ρ = 0.67 and 0.77, respectively).
Conclusions
Increased strain is associated with a proinflammatory lung response in patients with ALI.
Similar content being viewed by others
Introduction
There is substantial evidence that mechanical ventilation can cause some degree of injury to the lungs. In experimental models, this relationship is straightforward and has been termed ventilator-induced lung injury [1]. High tidal volumes or transpulmonary pressures applied to intact or pre-injured lungs can damage the parenchyma and distal organs. In patients, however, these relationships are sometimes confounded by the existence of previous injuries and the application of less aggressive ventilatory patterns. Nevertheless, it has been demonstrated that protective strategies focused on ameliorating the ventilator-induced lung response (called ventilator-associated lung injury, VALI [2]), lead to an improved prognosis in patients with acute lung injury (ALI) [3].
Positive-pressure ventilation can trigger a biological response within the lungs [4, 5]. From a physical point of view, it was recently proposed that both the force applied to the lungs (stress) and the magnitude of lung deformation (strain, ratio between tidal and end-expiratory lung volumes) are key concepts to explain the origin of this response [6]. Gattinoni et al. [7] hypothesized that lung injury occurs only whenever both stress and strain go beyond a certain threshold value. Although multiple pathways may be involved in lung damage, extracellular matrix remodeling [8] and an exacerbated proinflammatory response [9] have been described as relevant pathogenetic mechanisms in this scenario.
The relationships between respiratory mechanics and lung biological responses have been addressed in some studies [10–12]; recently, high strain levels have been associated with ventilator-induced lung injury [13]. Moreover, the metabolic activity of lung tissue appeared to be correlated with regional strain [14]. Our hypothesis was that the biological response, in terms of matrix remodeling and inflammation, should be proportional to the degree of lung strain. To test this hypothesis, we tried to correlate the lung strain due to the ventilatory settings employed with matrix remodeling and inflammation biomarkers in a group of patients with ALI under mechanical ventilation. Ventilated patients with normal lung function were used as controls.
Methods
Patients
Patients with more than 24 h of constant-flow, volume-controlled ventilation (Engstrom Carestation, General Electric Healthcare, USA) and expected need for ventilatory support for more than 2 days were included in the study. Ventilatory parameters were set by the responsible physician and kept constant throughout the study. Exclusion criteria were age below 18 years old, more than 5 days of mechanical ventilation, FiO2 greater than 80% (a lower FiO2 level is needed to perform an accurate measurement of EELV, see below), hemodynamic instability unresponsive to vasoactive drugs, immunosuppression, presence of air leaks or contraindications to bronchoalveolar lavage. Patients with acute lung diseases not meeting ALI criteria (e.g., unilobar pneumonia with respiratory failure) were also excluded. All patients were receiving sedation and analgesia as set by the attending physician. None of them had suspected or documented intra-abdominal hypertension. The regional ethics committee approved the protocol and informed consent was obtained from each patient’s next of kin.
Study protocol
After inclusion, demographic and clinical data (age, weight, height, diagnoses, APACHE-II score [15] and lung injury score (LIS) [16]) were collected; an arterial blood sample was drawn for PaO2 and PaCO2 measurement (GEM4000, Instrumentation Laboratory, USA), and ventilatory settings were recorded (including end-inspiratory and end-expiratory pressures after a 3-s hold). ALI was defined according to the American–European consensus conference criteria [17]. Respiratory system compliance was calculated as the ratio between tidal volume and driving pressure (plateau pressure minus PEEP). Afterwards, end-expiratory lung volume (EELV) was measured with an oxygen washin/washout technique (FRC INView, GE Healthcare, USA). Briefly, EELV was computed by estimating the change in nitrogen volume after a 20% increase in FiO2 [18, 19]. A second EELV measurement was obtained after returning FiO2 to the baseline value. The correlation between both EELV values was good (mean difference between consecutive measurements = −126 ± 162 ml; intraclass correlation coefficient for absolute agreement = 0.975) and they were subsequently averaged. Lung strain was computed as the ratio between tidal volume and EELV. Finally, a bronchoalveolar lavage fluid (BALF) sample was obtained. A 61-cm-long catheter (Gentle-Flo, Tyco Heathcare, Ireland) was inserted into the endotracheal tube until the tip of the catheter emerged beyond the distal end of the tube and placed in wedge position. The lung was lavaged with up to three 20-ml aliquots of sterile saline. An aliquot was cultured to document the presence of bacteria using standard microbiological techniques. A bacterial count higher than 104 CFU/ml was considered as positive. The remaining volume was filtered through sterile gauze, centrifuged at 1,500 rpm for 15 min to remove cells, and the supernatants were stored at −80°C for subsequent analysis.
Biochemical measurements
The following extracellular molecules were measured in BALF. As markers of extracellular matrix remodeling [20], matrix metalloproteinases (MMP) -2 and -9 were quantified by gelatin zymography as described elsewhere [21]. Additionally, the amino-terminal type-III procollagen peptide was measured by radioimmunoassay (UniQ PIIINP assay, Orion Diagnostica, Finland) as a marker of collagen synthesis. Tissue inhibitor of metalloproteases-1 (TIMP-1) was also measured (Flowcytomix kit, Bender Medsystems, Austria). The ratio MMP-9/TIMP-1 was computed as a marker of the proteolytic activity in the lung [22].
As lung injury may result in a disturbance in the equilibrium between pro- and anti-inflammatory mediators [10, 12], both pro- (IFNγ, IL-6, IL-8, IL-17A) and anti-inflammatory (IL-10) cytokines were measured in BALF using a multiplexing technique (Flowcytomix kit, Bender Medsystems, Austria) in a flow cytometer (Cellular Cytomics FC500, Beckman Coulter, USA). Finally, the growth factor VEGF, which may be involved in lung injury [23], was also measured using this technique.
Statistics
Data are presented as mean ± standard deviation. Variables were compared using a Mann–Whitney test (for comparisons between two groups) or a Kruskal–Wallis test (for three groups). Post hoc tests were performed using a Mann–Whitney test and p values were modified using the Bonferroni correction when appropriate. Bivariate correlations were computed using the Spearman coefficient. A p value lower than 0.05 was considered significant.
Results
Patients
Twenty-two patients were included in the study; sixteen of them met ALI criteria and the other six had healthy lungs. Detailed demographic and clinical data are presented in Table 1.
EELV and strain
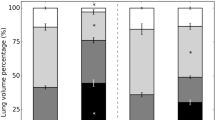
There were no differences in tidal volume between control and ALI patients. As expected, ALI patients had a lower EELV than controls, either in absolute values or after normalization by height. This difference resulted in higher strain levels in ALI patients than in controls (Table 1). The median strain value in the ALI group was 0.27. All controls had a strain value below this number. Therefore, this value was chosen as cutoff point to split the ALI group between patients with “normal strain” and those with “high strain”. Figure 1 shows the distribution of strain values among the three groups. Mean strain values are presented in Table 2.
Lung injury score, gas exchange, and respiratory mechanics
These results are shown in Table 2. Unsurprisingly, lung injury scores were higher in patients meeting ALI criteria. Similarly, control patients had higher PaO2/FiO2 ratios than those with ALI. The difference in PaO2/FiO2 between normal and high strain ALI patients was not significant (p = 0.28 in post hoc test). There were no differences in PaCO2 among groups. The number of patients with positive BALF cultures did not differ among groups.
Patients with ALI showed higher plateau and end-expiratory pressures than controls, with no differences in tidal volumes. Again, there were no differences between the normal and high strain subgroups. Driving pressures were higher in patients with ALI and high strain, although the differences with those with normal strain were not significant (p = 0.15 in post hoc test).
As expected, patients included in the high strain group showed higher values for this parameter and lower EELV values. There were no differences between control and ALI–low strain groups regarding these two parameters.
Biological response within the lungs
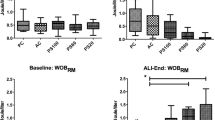
Regarding extracellular matrix remodeling, there were no significant differences in MMP-9 (p = 0.85), TIMP-1 (p = 0.61), the MMP-9/TIMP-1 ratio (p = 0.62), or collagen (p = 0.97) among groups (Fig. 2). MMP-2 signal was very weak and, therefore, it was not quantified. IFNγ and IL-17A were detected only in one patient. On the other hand, there were significant differences in proinflammatory cytokines IL-6 and IL-8 (p = 0.008 and p = 0.004 in Kruskal–Wallis tests, respectively). Patients with ALI and high strain showed increased levels of IL-6 and IL-8 in BALF, with no differences between controls and the normal strain ALI group (Fig. 3a, b). There were no differences in IL-10 (p = 0.18, Fig. 3c) or VEGF (p = 0.28, Fig. 3d).
Bivariate regression analysis showed a significant correlation between strain and levels of IL-6 (Spearman’s coefficient ρ = 0.67, p = 0.003) and IL-8 (ρ = 0.77, p < 0.001) in BALF (Fig. 4). There were no significant correlations of any of the measured molecules with other variables derived from lung mechanics (PEEP, plateau or driving pressures, or compliance).
Discussion
Our results demonstrate that there is a relationship between lung strain and alveolar proinflammatory response in patients with ALI, since patients with high strain show higher values of proinflammatory cytokines in BALF. In addition, we did not find any other physiological or clinical parameter with a similar correlation with the inflammatory response.
The role of strain in VALI
Applying the concept of “strain” to mechanical ventilation and VALI was proposed by Gattinoni et al. [6, 7]. Strain represents lung deformation relative to its starting conditions and is proportionally related to the force needed to apply that deformation (stress, represented by transpulmonary pressure). Recently, it was proposed that lung deformation may be one of the key mechanisms of ventilator-induced lung injury [24].
There are no clear threshold values for tidal volume [25] or plateau pressure [26] that may ensure a safe ventilatory strategy. Thus, an approach based on stress and strain may have some advantages. The variability of FRC (and EELV) among patients with ALI [27, 28] results in different degrees of strain even if the tidal volume is the same. Similarly, the variability of chest wall elastance leads to different transpulmonary pressures for the same plateau pressure applied [29]. Our results show that a given group of ALI cases can be split into two subgroups, each with different biological responses, according to strain. This is in agreement with a recent study by Bellani et al. [14] showing that the metabolic activity within the normally aerated lung increases with regional strain.
One of the key findings of this study is the absence of significant differences in clinical or mechanical parameters other than strain among the three subgroups. Although it is possible that the study may be underpowered to detect significant differences in gas exchange, plateau or driving pressures, there is a substantial overlapping of these parameters’ values in the three groups. Moreover, none of the classical variables related to lung mechanics correlated with the immune response in BALF.
Therefore, strain measurement could be a useful tool in the identification of ventilated patients at risk of exacerbated lung inflammation. Both IL-6 and IL-8 have a proinflammatory role and have been involved in the majority of studies dealing with VALI [12, 30]. It has been demonstrated that these two molecules are abundantly present in patients with ALI not receiving a low tidal volume ventilation, and that they have prognostic value for death and ventilator-free days [31].
Strain measurements
In their seminal study [6], Gattinoni et al. computed strain as the ratio of tidal volume to functional residual capacity (i.e., at zero end-expiratory pressure, ZEEP). In patients with PEEP, the increase in volume induced by the expiratory pressure is added to the tidal volume. If calculated this way, all increases in PEEP would result in an increase in strain. As an alternative, we used EELV to calculate strain. Bellani and co-workers [14] estimated regional strain in an aerated lung following a similar approach and found a good correlation with the metabolic activity of the tissue. With this approach, every increase in PEEP will always decrease strain, as EELV increases. Using EELV this way allowed us to obtain measurements without removing PEEP. In patients with early phase ALI, when the injured lungs may be highly recruitable [32], decreasing PEEP to 0 cmH2O may lead to a significant impairment in gas exchange and respiratory mechanics.
In patients with ALI, PEEP has mixed effects: First, it induces recruitment, thus increasing “effective” FRC and therefore decreasing strain. Second, it increases the already aerated lung areas, thus increasing strain in those areas. Gattinoni’s method overestimates strain, as it does not take into account the effects of recruitment. Conversely, our end-inspiratory strain results in the underestimation of the lung strain caused by the increase from FRC to EELV. As recruitment is highly variable in patients with ALI [33], the optimal strategy to calculate strain in patients with ALI and PEEP is not clear.
The method used to compute strain can also explain the difference in threshold values. Whereas Gattinoni et al. [13] proposed a strain value above 1.5–2 to be injurious, our value is quite lower. As a result of our experimental design, we cannot discard that other thresholds could yield different results, nor the existence of different thresholds for different biological responses. For instance, it has been hypothesized that the tissue deformation needed to induce matrix remodeling is higher than that required to induce an immune response [34].
We must admit that our results would be similar if EELV is used instead of strain. However, we cannot disallow the possibility that this is simply due to the lack of differences in tidal volume in our patients. Strain is a more general concept, so we chose to use it as the grouping factor. The contribution of tidal volume and EELV to strain and its consequences warrants further research.
A link between strain and the biological response
The biological lung response to mechanical ventilation features, among other manifestations, an increase of the extracellular matrix turnover and an immune response. Regarding the extracellular matrix, we did not find any differences in the levels of procollagen, MMP-2 or -9, or TIMP-1. Other authors found a significant increase in some of these molecules in patients with ALI [20]. Demoule et al. [11] found that the subset of patients with very low compliance (below 28 ml/cmH2O) featured increased levels of procollagen and MMP-2. The lack of differences in these parameters in the present study may be to do with the fact that most of our patients’ compliance values were above that threshold.
The relationship between lung strain and immune response can be explained by two different mechanisms. First, we can assume that the sickest patients have a more severe lung injury, which may result in a decrease of EELV and an increased release of inflammatory cytokines. If this were the case, strain could then be an independent marker of severity in patients with ALI. Although we cannot reject this argument, we did not find any clinical difference in terms of severity scores, gas exchange or lung mechanics between the two groups of ALI patients.
Alternatively, strain has been proposed to be one of the major determinants of VALI. In this framework, lung deformation beyond a certain threshold may lead to a mechanotransduction process that ultimately results in the release of proinflammatory cytokines. Zones at the interfaces between well-aerated and poorly aerated lung regions (i.e., where a higher deformation can occur) are at special risk of VALI. Therefore, it is possible that patients with high intratidal recruitment, who have increased severity and risk of death, will also show an increased overall strain [35]. However, it was recently shown that lung zones undergoing cyclic changes in aeration do not show an increased metabolic activity [14]. The underlying mechanism linking regional tissue deformation, overall strain, and inflammatory response remains to be clarified.
In spite of being global parameters, a high EELV and low strain could suggest that the deformation induced by tidal volume is relatively small and well tolerated. The correlation between strain and cytokines could support this hypothesis. However, the limited sample size of the study precludes any firm conclusion (especially in subgroups of patients), and the relationship between strain and cytokines is no proof of causality.
Irrespective of whether increased strain is the cause or the consequence of the observed proinflammatory response, our data suggest that strain could help in the identification of high-risk patients. There is increasing evidence that the most severe cases of ALI may benefit from more aggressive treatments, such as high PEEP levels or prone ventilation. The potentially helpful role of strain in titrate therapies for ALI is yet to be determined.
Conclusions
Our results show that patients with ALI and high values of strain show an increased alveolar inflammatory response. Moreover, we did not find any parameter other than strain or EELV that could help to identify this subset of patients. Therefore, we conclude that strain measurement may be a relevant parameter to address the biological lung response to injury and mechanical ventilation.
References
Tremblay LN, Slutsky AS (2006) Ventilator-induced lung injury: from the bench to the bedside. Intensive Care Med 32:24–33
American Thoracic Society, European Society of Intensive Care Medicine, Societe de Reanimation Langue Francaise (1999) International consensus conferences in intensive care medicine. Ventilator-associated lung injury in ARDS. Intensive Care Med 25:1444–1452
Petrucci N, Iacovelli W (2007) Lung protective ventilation strategy for the acute respiratory distress syndrome. Cochrane Database Syst Rev CD003844
Plataki M, Hubmayr RD (2010) The physical basis of ventilator-induced lung injury. Expert Rev Respir Med 4:373–385
dos Santos CC, Slutsky AS (2006) The contribution of biophysical lung injury to the development of biotrauma. Annu Rev Physiol 68:585–618
Chiumello D, Carlesso E, Cadringher P, Caironi P, Valenza F, Polli F, Tallarini F, Cozzi P, Cressoni M, Colombo A, Marini JJ, Gattinoni L (2008) Lung stress and strain during mechanical ventilation for acute respiratory distress syndrome. Am J Respir Crit Care Med 178:346–355
Gattinoni L, Protti A, Caironi P, Carlesso E (2010) Ventilator-induced lung injury: the anatomical and physiological framework. Crit Care Med 38:S539–S548
Pelosi P, Rocco PR (2008) Effects of mechanical ventilation on the extracellular matrix. Intensive Care Med 34:631–639
Pedreira PR, Garcia-Prieto E, Albaiceta GM, Taboada F (2006) Inflammatory response and apoptosis in acute pulmonary injury. Med Intensiva 30:268–275
Ranieri VM, Suter PM, Tortorella C, De Tullio R, Dayer JM, Brienza A, Bruno F, Slutsky AS (1999) Effect of mechanical ventilation on inflammatory mediators in patients with acute respiratory distress syndrome: a randomized controlled trial. JAMA 282:54–61
Demoule A, Decailliot F, Jonson B, Christov C, Maitre B, Touqui L, Brochard L, Delclaux C (2006) Relationship between pressure-volume curve and markers for collagen turn-over in early acute respiratory distress syndrome. Intensive Care Med 32:413–420
Stuber F, Wrigge H, Schroeder S, Wetegrove S, Zinserling J, Hoeft A, Putensen C (2002) Kinetic and reversibility of mechanical ventilation-associated pulmonary and systemic inflammatory response in patients with acute lung injury. Intensive Care Med 28:834–841
Protti A, Cressoni M, Santini A, Langer T, Mietto C, Febres D, Chierichetti M, Coppola S, Conte G, Gatti S, Leopardi O, Masson S, Lombardi L, Lazzerini M, Rampoldi E, Cadringher P, Gattinoni L (2011) Lung stress and strain during mechanical ventilation: any safe threshold? Am J Respir Crit Care Med 183:1354–1362
Bellani G, Guerra L, Musch G, Zanella A, Patroniti N, Mauri T, Messa C, Pesenti A (2011) Lung regional metabolic activity and gas volume changes induced by tidal ventilation in patients with acute lung injury. Am J Respir Crit Care Med 183:1193–1199
Knaus WA, Draper EA, Wagner DP, Zimmerman JE (1985) APACHE II: a severity of disease classification system. Crit Care Med 13:818–829
Murray JF, Matthay MA, Luce JM, Flick MR (1988) An expanded definition of the adult respiratory distress syndrome. Am Rev Respir Dis 138:720–723
Bernard GR, Artigas A, Brigham KL, Carlet J, Falke K, Hudson L, Lamy M, LeGall JR, Morris A, Spragg R (1994) Report of the American–European consensus conference on ARDS: definitions, mechanisms, relevant outcomes and clinical trial coordination. The Consensus Committee. Intensive Care Med 20:225–232
Olegard C, Sondergaard S, Houltz E, Lundin S, Stenqvist O (2005) Estimation of functional residual capacity at the bedside using standard monitoring equipment: a modified nitrogen washout/washin technique requiring a small change of the inspired oxygen fraction. Anesth Analg 101:206–212
Chiumello D, Cressoni M, Chierichetti M, Tallarini F, Botticelli M, Berto V, Mietto C, Gattinoni L (2008) Nitrogen washout/washin, helium dilution and computed tomography in the assessment of end expiratory lung volume. Crit Care 12:R150
Ricou B, Nicod L, Lacraz S, Welgus HG, Suter PM, Dayer JM (1996) Matrix metalloproteinases and TIMP in acute respiratory distress syndrome. Am J Respir Crit Care Med 154:346–352
Albaiceta GM, Gutierrez-Fernandez A, Garcia-Prieto E, Puente XS, Parra D, Astudillo A, Campestre C, Cabrera S, Gonzalez-Lopez A, Fueyo A, Taboada F, Lopez-Otin C (2010) Absence or inhibition of matrix metalloproteinase-8 decreases ventilator-induced lung injury. Am J Respir Cell Mol Biol 43:555–563
Lanchou J, Corbel M, Tanguy M, Germain N, Boichot E, Theret N, Clement B, Lagente V, Malledant Y (2003) Imbalance between matrix metalloproteinases (MMP-9 and MMP-2) and tissue inhibitors of metalloproteinases (TIMP-1 and TIMP-2) in acute respiratory distress syndrome patients. Crit Care Med 31:536–542
Abadie Y, Bregeon F, Papazian L, Lange F, Chailley-Heu B, Thomas P, Duvaldestin P, Adnot S, Maitre B, Delclaux C (2005) Decreased VEGF concentration in lung tissue and vascular injury during ARDS. Eur Respir J 25:139–146
Albaiceta GM, Blanch L (2011) Beyond volutrauma in ARDS: the critical role of lung tissue deformation. Crit Care 15:304
Deans KJ, Minneci PC, Cui X, Banks SM, Natanson C, Eichacker PQ (2005) Mechanical ventilation in ARDS: one size does not fit all. Crit Care Med 33:1141–1143
Hager DN, Krishnan JA, Hayden DL, Brower RG (2005) Tidal volume reduction in patients with acute lung injury when plateau pressures are not high. Am J Respir Crit Care Med 172:1241–1245
Ibanez J, Raurich JM, Moris SG (1982) A simple method for measuring the effect of PEEP on functional residual capacity during mechanical ventilation. Crit Care Med 10:332–334
Macnaughton PD, Evans TW (1994) Measurement of lung volume and DLCO in acute respiratory failure. Am J Respir Crit Care Med 150:770–775
Talmor D, Sarge T, Malhotra A, O’Donnell CR, Ritz R, Lisbon A, Novack V, Loring SH (2008) Mechanical ventilation guided by esophageal pressure in acute lung injury. N Engl J Med 359:2095–2104
Jorens PG, Van Damme J, De Backer W, Bossaert L, De Jongh RF, Herman AG, Rampart M (1992) Interleukin 8 (IL-8) in the bronchoalveolar lavage fluid from patients with the adult respiratory distress syndrome (ARDS) and patients at risk for ARDS. Cytokine 4:592–597
Parsons PE, Eisner MD, Thompson BT, Matthay MA, Ancukiewicz M, Bernard GR, Wheeler AP (2005) Lower tidal volume ventilation and plasma cytokine markers of inflammation in patients with acute lung injury. Crit Care Med 33:1–6
Albaiceta GM, Taboada F, Parra D, Luyando LH, Calvo J, Menendez R, Otero J (2004) Tomographic study of the inflection points of the pressure–volume curve in acute lung injury. Am J Respir Crit Care Med 170:1066–1072
Gattinoni L, Caironi P, Cressoni M, Chiumello D, Ranieri VM, Quintel M, Russo S, Patroniti N, Cornejo R, Bugedo G (2006) Lung recruitment in patients with the acute respiratory distress syndrome. N Engl J Med 354:1775–1786
Gattinoni L, Carlesso E, Cadringher P, Valenza F, Vagginelli F, Chiumello D (2003) Physical and biological triggers of ventilator-induced lung injury and its prevention. Eur Respir J Suppl 47:15s–25s
Caironi P, Cressoni M, Chiumello D, Ranieri M, Quintel M, Russo SG, Cornejo R, Bugedo G, Carlesso E, Russo R, Caspani L, Gattinoni L (2009) Lung opening and closing during ventilation of acute respiratory distress syndrome. Am J Respir Crit Care Med 181:578–586
Acknowledgments
Study supported by a Grant from General Electric Healthcare and by FICYT (COF-08-20 and COF-11-40). AGL is the recipient of a grant from Universidad de Oviedo (UNOV-09-BECDOC). IUOPA is supported by Obra Social Cajastur. CIBER-Enfermedades Respiratorias is supported by Instituto de Salud Carlos III. The authors would like to thank Ana Salas and Estela Rodríguez for their invaluable help with cytokine measurements, and Pablo Martinez Camblor for his help with the statistical analysis.
Author information
Authors and Affiliations
Corresponding author
Additional information
A. González-López and E. García-Prieto contributed equally to this work.
Rights and permissions
About this article
Cite this article
González-López, A., García-Prieto, E., Batalla-Solís, E. et al. Lung strain and biological response in mechanically ventilated patients. Intensive Care Med 38, 240–247 (2012). https://doi.org/10.1007/s00134-011-2403-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-011-2403-1