Abstract
BACKGROUND: In patients with COPD, early detection and rapid treatment are essential to prevent its progression and exacerbations.
OBJECTIVE: To identify factors that delay COPD detection.
METHODS: We conducted a cross-sectional study of elderly COPD patients and healthy subjects in rural Japan. We measured respiratory and physical function (hand grip force, maximum inspiratory pressure, maximum expiratory pressure, total trace length, and toe grasp force), walking ability, and quality of life (QOL, measured with a visual analog scale). We conducted spirometry in 408 subjects, whom we divided into 2 groups: those whose ratio of FEV1 to forced vital capacity (FVC) was < 70% (the airway-obstruction group, n = 60), and those whose FEV1/FVC was ≥ 70% (the healthy-subjects group, n = 348). To avoid confounding, we then matched 60 subjects from the healthy-subjects group to the 60 in the airway-obstruction group for age, sex, height, and smoking history, so both groups had 60 subjects.
RESULTS: Physical function was significantly inferior in the airway-obstruction group. Variables reflecting instantaneous walking ability (maximum walking speed and walking time in the 10-m hurdle walk) were significantly inferior in the airway-obstruction group, but there were no significant differences in 6-min-walk test, which reflects walking endurance, or in QOL.
CONCLUSIONS: Walking endurance, QOL, and proximal muscle strength in the extremities of patients with COPD were well preserved, which prevented detection of COPD and hampered the subjects' motive for seeking medical care. In consequence, lack of awareness impeded the early detection of COPD. In primary healthcare for the general elderly population, spirometry is much easier to conduct than physical function tests, so we recommend that spirometry screening programs for early-stage COPD detection and staging.
- airway obstruction
- chronic obstructive pulmonary disease
- COPD
- elderly
- quality of life
- 6-minute walk test
- walking
- primary healthcare
Introduction
Initiating the treatment of COPD as soon as possible is recommended, utilizing smoking cessation, vaccination, pharmacotherapy, and avoidance of risk factors.1–3 In respiratory rehabilitation the prompt initiation of treatment is considered more effective with high-load endurance training4,5 that is ongoing6 and takes into consideration cost-benefit performance.7 In Japan, the Nippon COPD Epidemiology (NICE) study revealed that only a very small portion of patients with COPD are diagnosed with the disease, and that many people have undiagnosed COPD in the general population. In Japan, approximately 5.3 million individuals over age 40 are estimated to have COPD.8
Insidious and ordinary clinical symptoms (eg, chronic cough and chronic sputum) are often considered trivial by people with COPD, which delays COPD detection.9,10 Pinto-Plata et al assessed COPD patients' exercise performance according to the Global Initiative for Chronic Obstructive Lung Disease (GOLD) criteria and found no significant difference in exercise performance between the control group and the stage I COPD group; however, there were significant differences between the control group and the stage II through stage IV COPD groups. They concluded that detection of COPD patients via measurement of physical performance is difficult.11
The objective of the present study was to identify factors that delay COPD detection in the general elderly population. We measured respiratory and physical function, walking endurance (via 6-minute walk test) and instantaneous walking ability, exercise performance, quality of life (QOL), and ability to conduct activities of daily living (ADL) in elderly COPD patients and healthy subjects in rural Japan.
Methods
This cross-sectional study was approved by the ethics review board of Nishikyushu University. All subjects gave written informed consent after receiving verbal and written explanations that enrollment was voluntary, he/she would not suffer any disadvantage by not participating, and he/she was authorized to withdraw consent at any time.
Subjects
Between August 2008 and October 2009 we screened 634 elderly residents in a rural area of Fukuoka, Japan, who participated in a health promotion program. The screening included careful medical history, including smoking status, exposure to second-hand smoke, personal and family history of asthma, ADLs, and QOL. We excluded those who had a diagnosis of a respiratory disease (including respiratory tract infection); residents with medical or family history of asthma; residents who had painful diseases including joint disorders that impair gait; residents who had a severe medical complication; residents who had markedly decreased physical performance; and residents who were incapable of fully understanding the objective and methods of the study. We enrolled 437 subjects.
Withdrawals and Post-Testing Exclusions
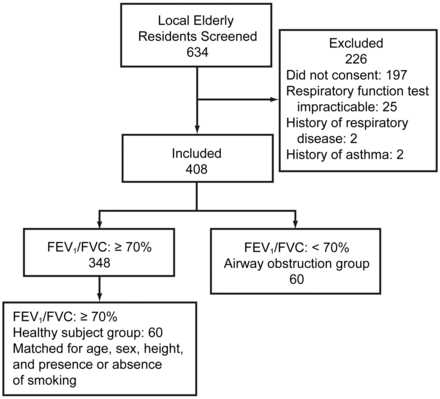
Of the 437 subjects who undertook spirometry, 29 had difficulty obtaining precise spirometry results, and they were excluded. The 408 remaining subjects were assigned to 2 groups: those whose ratio of FEV1 to forced vital capacity (FVC) was ≥ 70% (the healthy-subjects group, n = 348), and those whose FEV1/FVC < 70% (the airway-obstruction group, n = 60) (Fig. 1). To avoid confounding, we then matched 60 subjects from the healthy-subjects group to the 60 in the airway-obstruction group for age, sex, height, and smoking history, so both groups had 60 subjects. These 120 subjects and the study staff (other than the author who performed spirometry [JH]) did not know the subjects' group assignment. Table 1 shows the cohort's baseline characteristics.
Flow chart.
Baseline Characteristics of 120 Elderly Residents in Rural Japan
Cognitive Function Assessment
We assessed cognitive function with the Mini-Mental State Examination, which is a widely used simple assessment scale for cognitive state.12
Respiratory Function Testing
Spirometry (Autospiro AS507, Minato, Osaka, Japan) was conducted by a highly qualified pulmonary function technician (JH). The precision of the spirometry measurements was verified with the spirometer's software, which determined the forced expiration variables, and the flow-volume curves were printed. At least 2 spirometry maneuvers were performed, and the best value was recorded.
Respiratory Muscle Strength Assessment
We used a respiratory manometer (MicroRPM, Micro Medical, Kent, United Kingdom) to measure maximum inspiratory pressure (PImax) and maximum expiratory pressure (PEmax). PImax was measured at functional residual capacity or above, and PEmax at the maximum expiratory position or above. Two PEmax and PImax maneuvers were performed, and the best value was recorded.
Spinal Alignment Measurement
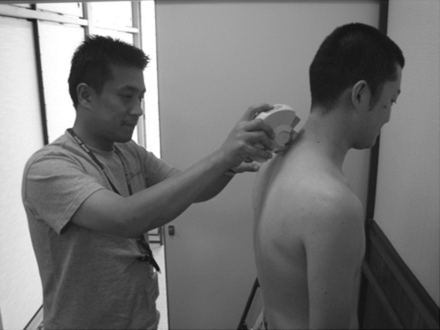
To measure spinal alignment we used a computerized spinal measurement tool (SpinalMouse, SpinalMouse Solutions/SG Fisher & Associates, Las Vegas, Nevada). The subject stands with open legs, and the paraspinal line is measured from the 7th cervical vertebra to the 3rd sacral vertebra; the total of angles formed between the upper and lower vertebral bodies of the first through 12th thoracic vertebrae are considered the thoracic kyphosis angle (Fig. 2). The total of angles formed between the upper and lower vertebral bodies of the first through 5th lumbar vertebrae are considered the lumbar lordosis angle.13 Three measurements were performed, and the mean value was used.
Assessment of spinal alignment.
Muscle Strength Assessment
To measure the muscle strength of the upper extremities we used a digital grip dynamometer (Grip-D, Takei Kiki Kogyo, Niigata, Japan) to measure hand grip force. To measure the strength of the quadriceps femoris we used a muscle strength gauge (μTAS, Anima, Tokyo, Japan) attached to a chair, with the subject in the sitting position, with the hip and knee joints bent at 90°. To measure the strength of the trunk, we counted the number of sit-ups the subject could do in 30 seconds. With the knees bent and both elbows crossed in front of the chest, each time the arms touched the right and left thighs and the subject's back touched the floor, it was counted as one sit-up.
We measured toe grasp force with a device (Yagami, Aichi, Japan) developed and studied by Murata et al (Fig. 3).14 Prior to each measurement we adjusted the device so that the distal phalanges of the great toe and fifth toe and the middle phalanges of the second to fifth toes could be placed on the toe grasp bar. With this device, the toes push the bar downward. Toe grasp force was measured twice, and the best value was recorded.
Assessment of toe grasp force.
Skeletal Muscle Mass Measurement
We used a 4-electrode dual-frequency body composition meter (DC-320, Tanita, Tokyo, Japan) to measure skeletal muscle mass via bioelectrical impedance analysis. The subject stood upright on the device. Skeletal muscle mass was measured once.
Balance Assessment
We used a stabilometer (GS-31, Anima, Tokyo, Japan) to assess balance. In the standing position, with eyes open, the subject was instructed: While looking at the mark (which was at a point 2 m away from the subject's eyes, at eye level), stand upright while closing your legs and bringing your arms against the sides of your body. This measurement lasted for 30 seconds, and the total length of the centroid tracing was assessed twice. One-leg standing time was measured twice for the right leg and the left leg, with the same procedure as for assessment of body sway. The best value was recorded.
Walking Ability Assessment
The subject was instructed to walk at his or her maximum walking speed on a flat floor, 11 m long. We used a stopwatch to measure the time required for the subject to pass the halfway point (5 m from the start point), and calculated the maximum walking speed. In the 10-m hurdle walk, 6 sponge hurdles (20 cm tall) were placed at 2-m intervals on a 10-m straight line, and we measured the time required to walk the hurdle course. In the timed up-and-go test15,16 we measured the time required to arise from the seated position, walk to a target placed 3 m away from the chair, and return to the seated position. In the 6-minute walk test the subject walked as far as possible in 6 min. We conducted the 6-minute walk test once. The other tests were performed twice, and we recorded the best value.
Quality of Life Assessment
We used a visual analog scale to assess the subject's views on 4 QOL domains: sense of well-being, life satisfaction level, purpose of life, and human relationships. The subject indicated his or her subjective assessment on a 100-mm straight line, on which zero indicated the most negative response and 100 indicated the most positive response.17–20 To assess performance of activities of daily living we used the Tokyo Metropolitan Institute of Gerontology questionnaire on activities of daily living (Table 2). The score was the total number of yes responses.
Questionnaire on Activities of Daily Living*
Statistical Analysis
With statistics software (SPSS 17.0, SPSS Japan, Tokyo, Japan) we analyzed the data from the healthy-subjects group and the airway-obstruction group with the paired t test. Values are expressed as mean ± SD. A P value < .05 was considered statistically significant.
Results
Symptoms
The only symptom we observed was chronic sputum in 2 patients in the airway-obstruction group who had stage II COPD. All other patients in the airway-obstruction group had no symptoms (chronic cough, chronic sputum, or exertional dyspnea).
Prevalence and Severity of COPD
In 60 of the 408 subjects who could perform the respiratory function test, the FEV1/FVC was < 70%. The prevalence of COPD was 15%. The GOLD COPD severity distribution was: stage I (16 patients, 27%); stage II (33 patients, 55%); stage III (11 patients, 18%); and stage IV (no patients). Therefore, patients with mild (stage I) to severe (stage III) COPD were successfully detected.
Comparisons of Healthy-Subjects Group and Airway-Obstruction Group
Table 1 shows the anthropometry, spirometry, Mini-Mental State Examination, and trail-making test data. The airway-obstruction group had significantly lower body mass index, FEV1, FEV1/FVC, and Mini-Mental State Examination score. There were no significant differences between the groups in FVC, percent-of-predicted FVC, or trail-making test score.
Table 3 shows the strength, exercise, and walking performance results. The airway-obstruction group had significantly lower hand grip force, PImax, PEmax, instantaneous walking ability (ie, maximum walking speed), and 10-m hurdle walk time. There was no significant difference in the other measured variables. Table 4 shows the QOL and ADL results. There were no significant differences in QOL, but the ADL values were significantly higher in the healthy-subjects group.
Strength, Exercise, and Walking Performance*
Subjective Quality of Life and Activities of Daily Living Scores*
Discussion
In clinical settings we frequently encounter patients with moderate to severe COPD, but it is difficult to provide them with effective exercise training and to improve their exercise performance. In the present study we did not make a differential diagnosis from other disorders (eg, asthma or respiratory infection) because we had excluded people with diagnosed respiratory diseases. Our study suggests that spirometry in elderly subjects can detect undiagnosed COPD. Currently, primary care physicians insufficiently recognize the importance of spirometry for early COPD detection,21,22 so programs are needed to enable professionals to precisely perform spirometry.23,24 Technically, it is not difficult to perform spirometry in communities where many elderly individuals reside, so screening spirometry has a potentially great contribution to early COPD detection.
Walking endurance was well preserved in our subjects, as indicated by no significant difference between the groups in the 6-minute walk test. Furthermore, none of the subjects complained of a decrease in QOL or ability to do their ADLs, which caused them to be unaware of their COPD. Pinto-Plata et al11 found no significant difference in exercise performance between stage I COPD patients and healthy subjects. Therefore, we think it is difficult for COPD patients to notice the disease through decreased exercise performance. The proportion of muscle fibers, especially type I fibers, has been reported to decrease in COPD patients, compared to healthy elderly subjects,25–27 and the decrease is associated with the severity of COPD.25,27 We speculate that the relevant decrease is not as intense in early-stage COPD patients, which makes it more difficult for them to notice a decrease in walking endurance from muscle fatigue or other clinical manifestations, compared to healthy subjects.
Furthermore, 4–35% of COPD patients have muscular atrophy.28–30 Atrophy of the quadriceps femoris reduces exercise performance31,32 and QOL.33 However, we found no significant difference in quadriceps femoris strength or muscle mass, so we think there was no marked muscular atrophy in our airway-obstruction group, which probably also made them less sensitive to decreases in walking endurance and QOL.
Walking endurance and QOL in our COPD patients were well preserved, although there was a significant difference in instantaneous walking ability between the healthy and airway-obstruction subjects. Furthermore, muscle strength was significantly less in the distal than the proximal muscles in the extremities. We suspect that these are the primary factors that delay COPD diagnosis, because the patients are unaware or less aware of the disease, which hampers their motivation for seeking medical care and delays COPD detection.
Our data also indicate that instantaneous walking ability, muscle strength in the distal extremities, and respiratory muscle strength had started to decline in the airway-obstruction subjects, despite that few of them had symptoms (eg, chronic cough, chronic sputum, or exertional dyspnea). Decreased distal muscle strength in the extremities involves many points to be elucidated. COPD patients with decreased exercise performance have been reported to have well preserved hand grip force, similar to healthy subjects.34 However, most previous studies on muscular functions in COPD patients examined the quadriceps femoris and the diaphragm, and few studies have examined distal muscle strength in the extremities. Our data suggest that a decrease in muscle strength occurs first in the distal muscles of the extremities. We presume that initially decreased strength in small distal muscles of the extremities is less likely to impair the person's ability to do ADLs, so the impairment is not highly noticeable.
Regarding respiratory muscle strength, on the other hand, there were significant differences in PImax and PEmax between the airway-obstruction and healthy-subjects groups. Changes in muscle fibers have been reported to occur earlier in the diaphragm than in the quadriceps femoris.35 Furthermore, patients with mild to moderate COPD have atrophy of the respiratory muscles.36 These facts explain the significant difference we found in respiratory muscle strength between the airway-obstruction and healthy-subjects groups.
Ofir et al37 reported that the effects of the abnormal dynamic ventilatory system are already manifested in patients with stage I COPD. Dynamic hyperinflation causes exertional dyspnea,38 but in early-stage COPD the hyperinflation-induced effects on the diaphragm and intercostal respiratory muscles readily occur, rather than subjective symptoms, which possibly leads to decreases in respiratory muscle strength. Previous studies have reported that, in patients with severe COPD, respiratory muscle strength is significantly related to exercise performance and exertional dyspnea,39–41 ADL performance,42 and QOL.39,43 We conjecture that the abnormalities in the dynamic ventilatory system in our airway-obstruction group were manifested not as subjective symptoms but as decreases in respiratory muscle strength, which made ADL performance impairment more difficult to detect.
Many studies have demonstrated the clinical efficacy of exercise training programs for patients with respiratory disease.4–6 We think pulmonary rehabilitation programs for COPD patients should include resistance training for the distal muscles of the extremities,44 the respiratory muscles, and instantaneous walking ability.
In consideration of the fact that respiratory muscle strength started to decrease in COPD patients, as manifested by decreased PImax and PEmax, we speculate that prompt implementation of respiratory muscle training might delay decreases in ADL performance and QOL. There is moderate evidence of the benefits of such exercise programs on exercise performance,39–41 ADL performance,42 and QOL.39,43 Furthermore, respiratory muscle training programs and instantaneous walking ability are clinically relevant when determining the composition of respiratory rehabilitation programs in medical care.
Limitations
First, we did not perform a bronchodilator test to make a differential diagnosis of COPD. However, we did take careful personal and family medical histories and excluded subjects with known asthma. But it is possible that we included some patients with asthma in the airway-obstruction group. Second, the study population was elderly subjects who participated in a health promotion program, so they had high consciousness about their health. Middle-age patients with COPD were excluded, so our study applies only to elderly subjects, not all ages of COPD patients in the general population. Third, we did not conduct a maximal exercise stress test, but, instead, the 6-minute walk test, which is a submaximal exercise stress test.
In the future we will conduct a prospective longitudinal study by following up the subjects in the present study's airway-obstruction group, and those patients will gain a definite diagnosis and undergo treatment (eg, pulmonary rehabilitation). We will also follow the natural course of COPD in a group without diagnosis and treatments. We will examine the time-course changes in their respiratory function, physical performance, and QOL.
Conclusions
Walking endurance, QOL, and proximal muscle strength in the extremities were well preserved in our COPD patients, although there was a significant difference in instantaneous walking ability between the healthy-subjects group and the airway-obstruction group. We suspect that the good preservation of these variables is the primary factor that delays COPD detection, because these individuals are unaware of the disease and therefore not motivated to seek medical care. In primary healthcare for the general elderly population, screening spirometry is needed to detect COPD, and we believe that health screening programs with spirometry should be implemented to detect and stage early COPD.
Acknowledgments
We thank Michie Ohyama, Welfare Equipment Net, for coordinating the study, and Satoshi Sakima MD for critical review of the manuscript.
Footnotes
- Correspondence: Jun Horie RPT MSc, Department of Rehabilitation, Nishikyushu University, Ozaki 4490–9, Kanzaki-cho, Kanzaki, Saga 842-8585, Japan. E-mail: horiej{at}nisikyu-u.ac.jp.
-
This study was partly supported by the Univers Foundation, Tokyo, Japan.
-
Mr Horie presented a version of this paper at the 45th Congress of the Japanese Physical Therapy Association, held May 28, 2010, in Gifu, Japan.
-
The authors have disclosed no conflicts of interest.
- Copyright © 2011 by Daedalus Enterprises Inc.