Abstract
BACKGROUND: The use of endoscopic ultrasound with bronchoscope-guided fine-needle aspiration (EUS-B-FNA) has been described in the evaluation of mediastinal lymphadenopathy. Herein, we conduct a meta-analysis to estimate the overall diagnostic yield and safety of EUS-B-FNA combined with endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA), in the diagnosis of mediastinal lymphadenopathy.
METHODS: The PubMed and EmBase databases were searched for studies reporting the outcomes of EUS-B-FNA in diagnosis of mediastinal lymphadenopathy. The study quality was assessed using the QualSyst tool. The yield of EBUS-TBNA alone and the combined procedure (EBUS-TBNA and EUS-B-FNA) were analyzed by calculating the sensitivity, specificity, positive likelihood ratio, negative likelihood ratio, and diagnostic odds ratio for each study, and pooling the study results using a random effects model. Heterogeneity and publication bias were assessed for individual outcomes. The additional diagnostic gain of EUS-B-FNA over EBUS-TBNA was calculated using proportion meta-analysis.
RESULTS: Our search yielded 10 studies (1,080 subjects with mediastinal lymphadenopathy). The sensitivity of the combined procedure was significantly higher than EBUS-TBNA alone (91% vs 80%, P = .004), in staging of lung cancer (4 studies, 465 subjects). The additional diagnostic gain of EUS-B-FNA over EBUS-TBNA was 7.6% in the diagnosis of mediastinal adenopathy. No serious complication of EUS-B-FNA procedure was reported. Clinical and statistical heterogeneity was present without any evidence of publication bias.
CONCLUSIONS: Combining EBUS-TBNA and EUS-B-FNA is an effective and safe method, superior to EBUS-TBNA alone, in the diagnosis of mediastinal lymphadenopathy. Good quality randomized controlled trials are required to confirm the results of this systematic review.
Introduction
Both endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) and endoscopic ultrasound-guided fine-needle aspiration (EUS-FNA) enable real-time aspiration of mediastinal lesions under direct vision.1–5 The 2 procedures have complementary access to the mediastinum.6–9 Although EBUS-TBNA provides an easy access to pretracheal and right paratracheal lesions, EUS-FNA is useful for accessing the inferior mediastinum, the left paratracheal area, and some areas of the aortopulmonary window. In a meta-analysis, the combination of the 2 procedures was found to provide greater sensitivity than either procedure alone in mediastinal staging of lung cancer.10 EBUS-TBNA and EUS-FNA are conventionally performed using a dedicated echobronchoscope and echoendoscope, respectively. In the former, the access to mediastinal lymph nodes is transtracheal or transbronchial, whereas the latter requires a transesophageal route. While EBUS-TBNA is generally performed by pulmonary physicians or thoracic surgeons, EUS-FNA is largely performed by gastroenterologists. This increases the cost, as well as waiting times, for patients requiring both procedures.
Hwangbo et al11 have reported the use of the echobronchoscope for carrying out transesophageal needle aspiration, termed as endoscopic ultrasound with bronchoscope-guided fine-needle aspiration (EUS-B-FNA). We have also described our initial experience with this technique recently in unselected subjects with mediastinal lymphadenopathy.12 In this study, we perform a systematic review and meta-analysis on the utility and safety of EUS-B-FNA in the diagnosis of mediastinal lymph node enlargement.
QUICK LOOK
Current knowledge
The combination of endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) and endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) provides greater sensitivity than either procedure alone. The use of the EBUS scope for performing EUS-FNA has been termed endoscopic ultrasound with bronchoscope-guided FNA (EUS-B-FNA).
What this paper contributes to our knowledge
The results of this study suggest that combining EUS-B-FNA with EBUS-TBNA is an effective and safe method, superior to EBUS-TBNA alone, in the diagnosis of mediastinal lymphadenopathy.
Methods
Search Strategy
We first searched the PubMed and EmBase databases for any systematic review on EUS-B-FNA; no such citation was found. Next, all the authors independently searched the PubMed and EmBase databases for relevant studies published between 2004 and May 2014 describing the diagnostic value of EUS-B-FNA in subjects with mediastinal lymphadenopathy using the following search terms: (ebus OR endobronchial ultrasound OR endobronchial ultrasonography OR eus OR echoendoscope OR endoscopic ultrasonography OR endoscopic ultrasound OR bronchoscopic ultrasound OR esophageal ultrasound OR ultrasound bronchoscope OR ultrasonic bronchoscope) AND (tbna OR tena OR needle aspiration OR fna). From the EmBase database, we included citations under only 2 categories: articles and articles in press. We reviewed the list of references of original studies, editorials, and reviews; and also sifted through our personal files. We excluded the following studies: (1) case reports, abstracts, comments, editorials, and reviews; (2) studies describing the combined use of EUS-FNA and EBUS-TBNA but not performed with the same echobronchoscope; (3) studies describing EUS-B-FNA in ≤ 20 subjects; (4) studies describing the transesophageal use of echoendoscope for sampling lesions other than those in the mediastinum.
Initial Review of Studies
The database thus created from the electronic searches was assimilated in the reference manager package Endnote X7 (Thomson Reuters, New York, New York), and all duplicate citations were discarded. Two authors (SD, RA) screened these citations by review of the title and abstract to identify the relevant studies. Any disagreement was resolved by discussion between the authors. This database was then scrutinized again to include only primary articles. The full text of each of these studies was obtained and reviewed in detail.
Data Abstraction
Data were entered into a standard data extraction form. The following items were extracted: (1) publication details (authors, year of publication, and other citation particulars including the country where the study was conducted); (2) study design (prospective or retrospective); (3) aim of the study, number of subjects, and inclusion criteria; (4) the nature of the operators (whether surgeons, pulmonary physicians, or gastroenterologists), positioning of the patient during the procedure, and the type of sedation used; (5) stations sampled, respective number of subjects and/or lymph nodes assessed by EBUS-TBNA and EUS-B-FNA; (6) size of lymph nodes on chest computed tomogram and/or EBUS and/or endoscopic ultrasound with an echobronchoscope (EUS-B); (7) diameter of EBUS-TBNA needle, number of passes made through EBUS/EUS-B, and availability of rapid on-site cytological examination; (8) the duration of the EBUS-TBNA and EUS-B-FNA procedures; (9) the sensitivity and specificity of EBUS-TBNA, EUS-B-FNA, and the combined procedure; (10) the additional yield of EUS-B-FNA, if reported; (11) reasons for performing EUS-B-FNA; and (12) complications associated with the procedure.
Assessment of Study Quality
The quality and validity of each study incorporated in this meta-analysis was assessed using the QualSyst tool for qualitative studies.13 This instrument is comprised of 10 questions each, with scores ranging from 0 to 2 and the highest total score being 20. Each article was independently adjudged by 2 authors (SD, RA) for the stated criteria. Weighted Cohen's kappa co-efficient was used to define the inter-observer agreement for selection of studies.
Statistical Analysis
The statistical software packages Meta-Disc 1.4 (Ramon Cajal Hospital, Barcelona, Spain) and StatsDirect 2.8.0 (StatsDirect, Cheshire, United Kingdom) were used to perform all the statistical analyses. The analyses performed in this study are on a per patient basis (and not per lymph node), and the test performance characteristics were derived from the raw data of each study.
Determination of the Pooled Effect
We analyzed the utility of EUS-B-FNA by calculating the sensitivity, specificity, positive likelihood ratio (PLR), negative LR (NLR), and diagnostic odds ratio (DOR; PLR/NLR) of individual studies for EBUS-TBNA alone and the combined procedure (EBUS-TBNA plus EUS-B-FNA). Sensitivity and specificity were pooled using the fixed effects model,14 whereas the PLR, NLR, and DOR were pooled using the DerSimonian-Laird random effects model to derive a pooled estimate with 95% CI.15,16
The additional diagnostic gain with EUS-B-FNA was analyzed by calculating the proportions for the individual studies (n/N, where n is the additional gain and N is the total number of subjects evaluated).17,18 The proportions were pooled using a DerSimonian random effects model, in the presence of significant heterogeneity.
Assessment of Heterogeneity
Heterogeneity for the individual outcomes was assessed using the I2 test, which measures the extent of inconsistency among the results of the studies. An I2 value ≥ 50% indicates significant heterogeneity.19 Heterogeneity was also assessed using the Cochran Q statistic, and a P value < 0.1 was considered significant.20
Estimation of Publication Bias
The presence of publication bias was evaluated using the funnel plot (log DOR on x-axis against standard error of DOR on y-axis).21 Publication bias was also investigated using the Egger test22 and the Begg-Mazumdar test.23 The DOR for the individual studies were calculated using the Meta-Disc software, and then these were entered into the StatsDirect package to construct the funnel plots.
An institutional review board clearance was not required for this study, as this was a meta-analysis of published studies.
Results
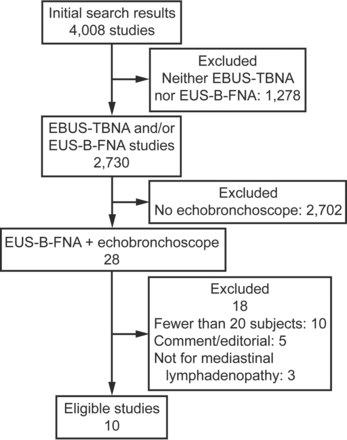
The initial database search retrieved a total of 4,008 citations, of which 10 studies (1,080 subjects) met our inclusion criteria (Fig. 1).11,24–32 All studies were observational; 6 were prospective,24,25,27,29,30,32 and 4 had a retrospective design (Table 1).11,26,28,31 Five studies were aimed at mediastinal staging of lung cancer,24–26,31,32 using a combination of EBUS-TBNA and EUS-B-FNA (Table 1). One study each was performed for the diagnosis of sarcoidosis, diagnosis of mediastinal lesions, molecular diagnosis of lung cancer, restaging of lung cancer after chemotherapy, and diagnosis of suspected malignant mediastinal lesions in those with non-diagnostic conventional techniques.11,27–30 The procedure was performed by pulmonologist(s) in 7 studies, a surgeon in one study, and both pulmonologists and surgeons in 2 studies (Table 2). The various nodal stations accessed by EBUS-TBNA or EUS-B-FNA are also listed in Table 2. The demonstration of malignancy by EBUS-TBNA and/or EUS-B-FNA was taken as true positive in all the studies, whereas surgical confirmation and/or follow-up was used in case of benign pathology (Table 3). A 22 gauge TBNA needle was used in most of the studies, a 21 gauge needle was used in a single study,29 and one study did not report the needle size.25 Rapid on-site cytological examination was not performed in any study. Of the 10 studies, the procedure was performed under conscious sedation in 8, general anesthesia in one, and either of the 2 modalities in one. The studies were generally of good quality (Table 4) with the median (interquartile range) score being 18 (18–19). The inter-observer agreement for scoring of study quality was good (weighted Cohen's kappa = 0.9).
Systematic review flow chart. EBUS = endobronchial ultrasound; EUS = endoscopic ultrasound; EUS-B-FNA = EUS with bronchoscope-guided fine needle aspiration.
Demographic Characteristics of Subjects in Studies Reporting the Performance of EUS-B-FNA for the Diagnosis of Mediastinal Lymphadenopathy
Details of the EBUS-TBNA and EUS-B-FNA Procedures in the Diagnosis of Mediastinal Lymphadenopathy
Criteria Used for Confirmation of Diagnosis in the Included Studies
QualSyst Tool for Assessment of Quality of the Included Studies
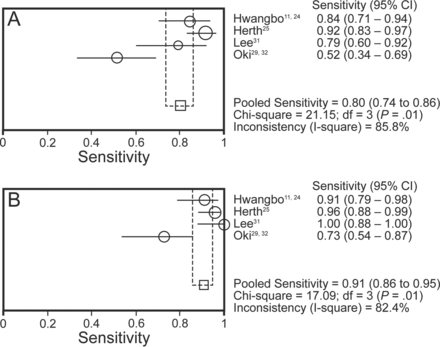
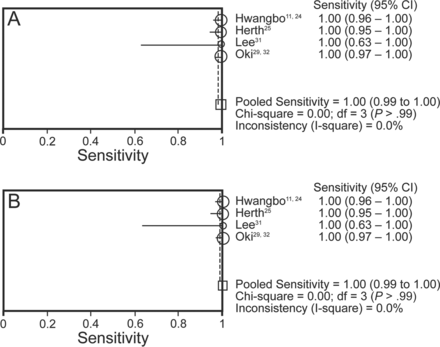
Four studies (465 subjects) provided data for the true and false positive, as well as the true and false negative, results of both EBUS-TBNA alone and the combined procedure, and were included in the diagnostic accuracy meta-analysis (Table 5).24,25,31,32 All these studies were aimed at mediastinal staging of lung cancer. The diagnostic sensitivity of EBUS-TBNA alone in these studies ranged from 52% to 92%, with the pooled sensitivity being 80% (95% CI 74–86%) by random effects model (Fig. 2). The diagnostic sensitivity of the combined procedure was 91% (95% CI 86–95%), and was significantly higher than the pooled sensitivity of EBUS-TBNA alone (P = .004). The number of combined procedures that need to be performed to achieve one additional diagnosis, as compared with EBUS-TBNA alone, is 10 (95% CI 6–29). The pooled specificity of EBUS-TBNA and the combined procedure was 100% (Fig. 3). The pooled sensitivity, specificity, PLR, NLR, and DOR are provided in Table 6.
Outcomes of the EBUS-TBNA, EUS-B-FNA, and Combined Procedures of the Included Studies
Forest plot of the summary sensitivity of A: endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) and B: the combined procedure in the diagnosis of mediastinal lymphadenopathy; df = degrees of freedom. The sensitivity of individual studies is represented by a circle, through which runs a horizontal line (95% CI). The square at the bottom represent the pooled sensitivity from the studies. There was a significant difference in the pooled sensitivity between the 2 groups (P = .004).
Forest plots of the summary specificity of A: endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) and B: the combined procedure in the diagnosis of mediastinal lymphadenopathy; df = degrees of freedom. The specificity of individual studies is represented by a circle, through which runs a horizontal line (95% CI). The square at the bottom represent the pooled specificity from the studies.
Summary Characteristics of EBUS-TBNA and the Combined Procedure (EBUS-TBNA and EUS-B-FNA)
Seven studies (653 subjects) reported data on the number of subjects in whom EUS-B-FNA achieved additional diagnostic yield over EBUS-TBNA.11,24,25,27,30–32 In one study, both EBUS-TBNA and EUS-B-FNA were not performed in all subjects; therefore, it was not included for calculating the additional diagnostic gain.28 The pooled additional diagnostic gain was 7.6% (95% CI 3.9–12.6%), as depicted in Figure 4. None of the studies reported any serious complication of the EUS-B-FNA procedure. One study reported the development of a lymph node abscess after EBUS-TBNA in one subject.24
Additional diagnostic gain of endoscopic ultrasound with bronchoscope-guided fine-needle aspiration (EUS-B-FNA) over endobronchial ultrasound-guided transbronchial needle aspiration (EBUS-TBNA) in subjects with mediastinal lymphadenopathy (random effects model). The gain in individual studies is represented by a circle (percentage) through which runs a horizontal line (95% CI). The square at the bottom represents the pooled additional diagnostic gain from the studies (7.6% [95% CI 3.9–12.6%]).
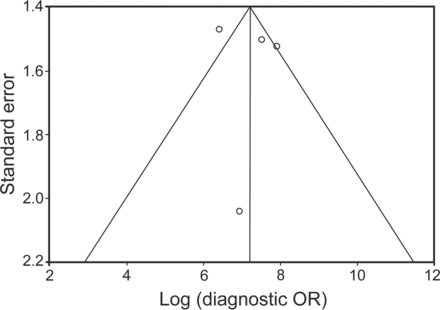
Clinical heterogeneity was evident in the nature of the studies (prospective vs retrospective), lymph nodes sampled, and number of aspirations per node (Tables 1 and 2). Significant statistical heterogeneity was also observed for the outcome of sensitivity of the combined procedure (I2 = 82.4%; Cochran Q statistic 17.09, P = .001). There was no evidence of publication bias on visual examination of the funnel plot (Fig. 5) or on statistical tests (Begg-Mazumdar: Kendall's tau = 0.3333, P = .75; Egger: bias = −0.2799, P = .91).
Funnel plot comparing diagnostic odds ratio (DOR) versus the standard error of DOR. Circles represent individual studies included in the meta-analysis. The vertical line in the center indicates the summary DOR. The other 2 lines represent the 95% CIs.
Discussion
The results of this systematic review suggest that the transesophageal use of the echobronchoscope is a safe and effective method of accessing the mediastinum, and provides incremental diagnostic yield over and above that achieved with EBUS-TBNA alone. Overall, we found a good incremental yield (approximately 8%) of adding EUS-B-FNA to the EBUS-TBNA procedure in the diagnosis of mediastinal lymphadenopathy. The sensitivity of the combined technique was significantly higher than EBUS-TBNA alone (91% vs 80%), in mediastinal staging of lung cancer. In fact, only 10 combined procedures need to be performed to achieve a diagnosis in one additional patient, when compared with EBUS-TBNA alone. The sensitivity of the combined technique is similar to the sensitivity (86%) reported in a meta-analysis of combined EBUS-TBNA plus EUS-FNA by Zhang et al10 However, our analysis is different from the previous meta-analysis in that we have included only those studies that have utilized the same echobronchoscope for performing both the transbronchial and the transesophageal procedures.
The transesophageal introduction of an echobronchoscope to access mediastinal lymph nodes was first described in 2007.33 Since then, this unconventional technique has been used not only for mediastinal staging of lung cancer but also for the diagnosis of sarcoidosis, molecular diagnosis of lung cancer, diagnosis of suspected malignant mediastinal lesions in those with non-diagnostic conventional techniques, restaging of lung cancer after chemotherapy, and others. The clinical importance of the combined technique lies in the fact that an additional benefit over EBUS-TBNA can be accomplished utilizing the same instrument in the same setting with the same operator as EBUS-TBNA. There are several other advantages of performing combined transbronchial and transesophageal needle aspiration procedure for mediastinal lymphadenopathy, using a single echobronchoscope (Table 7). It provides a wider access to the mediastinum than either procedure alone (stations 4L, 5, 8, and 9 better accessed with the transesophageal approach; stations 4R, 10, and 11 with the transbronchial approach). It potentially resolves many logistic difficulties by reducing the cost and wait times for patients. It also reduces the dependence of the pulmonary physician/thoracic surgeon on other clinical specialties. Moreover, the procedure is safe; no serious complication resulting from the EUS-B-FNA procedure was reported in any of the studies included in this review.
Individual Studies Reporting the Reasons for Performing EUS-B-FNA
The diagnostic sensitivity of EBUS-TBNA alone has been found to range from 78% to 92% in previous meta-analyses of studies involving subjects with sarcoidosis, lung cancer, or undiagnosed mediastinal lymphadenopathy.3,34,35 The pooled sensitivity of EBUS-TBNA alone in our study (80%) falls at the lower end of this range, possibly because rapid on-site cytological examination was not performed in any of the studies included in this review.
Finally, our analysis is not without limitations. The results of this analysis can be considered as hypothesis-generating, as none of the studies included in the analysis was a randomized controlled trial. This is likely to introduce bias in the selection of cases for EUS-B-FNA. There was also significant statistical heterogeneity and considerable clinical heterogeneity in this analysis because of the variations in the study design, inclusion criteria, primary objectives, different operators with varying expertise, the number of aspirations, and lymph node stations among the included studies. However, this clinical heterogeneity can also be considered as useful because it suggests that the combination of EUS-B-FNA and EBUS-TBNA is beneficial in different settings, which would reflect its effectiveness in real world situations. Further, as mentioned, none of the included studies employed rapid on-site cytological examination. The strengths include the inclusion of a large number of subjects and the use of robust statistical methods. The analysis also provides an estimate of the additional diagnostic benefit that an EBUS operator can accomplish by employing this effortless technique in selected patients with mediastinal lymph node enlargement.
Conclusions
Combining EUS-B-FNA with EBUS-TBNA was found to be a safe and effective method to increase the diagnostic yield in the evaluation of mediastinal lymphadenopathy. With most studies in this systematic review aimed at mediastinal staging of cancer, larger randomized trials from different centers, assessing the utility of this technique for other causes of mediastinal lymphadenopathy, are required to confirm the results of this meta-analysis.
Footnotes
- Correspondence: Ritesh Agarwal MD DM, Department of Pulmonary Medicine, Postgraduate Institute of Medical Education and Research, Sector 12, Chandigarh 160012, India. E-mail: agarwal.ritesh{at}live.com.
The authors have disclosed no conflicts of interest.
- Copyright © 2015 by Daedalus Enterprises